$OCC held an investor webinar this morning and it was clear from remarks from Chair John Van Der Wielen that the motivation for the session is their puzzlement at the SP slumping back below $1.00 following the recent peak ahead of the capital raise when it got up above $1.50. There was also a reference to commentary on some online platforms. (Of course, I assume they cannot be referring to the highly informed and expert analysis and opinions expressed on this honourable platform!)
For me the SP fall back is obvious. Although cashed up with over $50m providing a runway of likely a couple of years, $OCC is in that medical devices "valley of death" between initial US approval and initial sales, and investors lack clarity on the path to cashflow break even.
In that respect, I believe the company's lack of transparency as to details of revenue reporting is a contributory factor, and so I took the opportunity on the call to politely ask whether improved transparency might give investors better understanding and, therefore, confidence.
Management seem committed to increase the transparency via better "segment reporting", but I am not sure my recommendation of: Striate Global + Remplir ANZ + Remplir US + Remplir RoW will be taken onboard. The stated reason is of not wanting to reveal pricing insights for commercial confidential reasons. However, I am not convinced. One to keep chipping away at.
Rather than summarise the overall webinar, I want to zoom in on a few key messages and new information that was revealed in the Webinar.
1. US Progress
Management clearly wanted to explain the many stage gates you have to navigate in the US after FDA approval before sales start to flow, and I think they did a good job of this with the chart below. I believe this is not well-uinderstood by ASX investors who might not have followed mulitple medical devices through the US market.
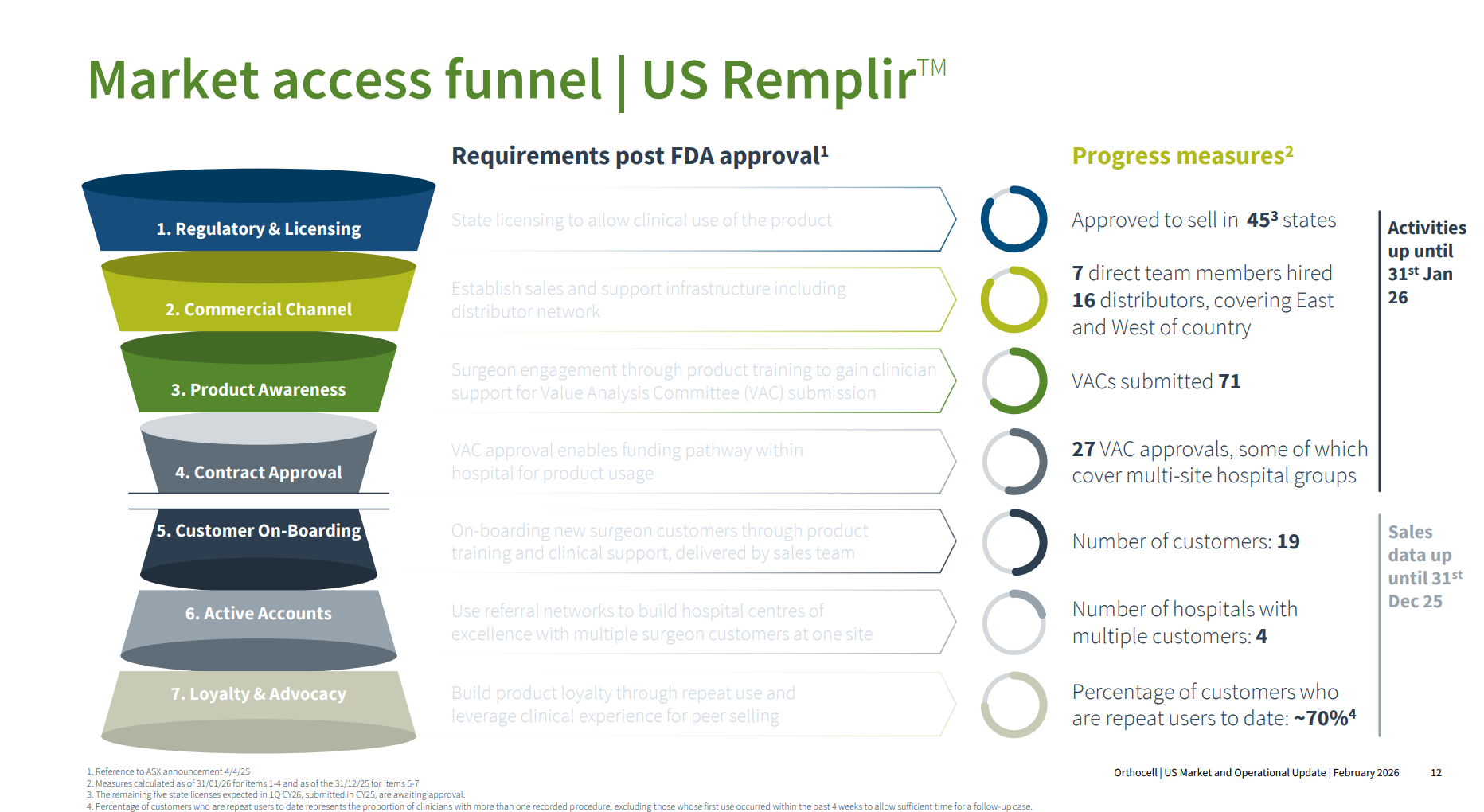
Importantly, some of the metrics are showing good movement between End-Dec and End-Jan as follows:
Distributors: From 14 to 16 ... with an ongoing process to expand coverage
VACs Sumitted: up from 61 to 71
VACs Approved: from 23 to 27, ... with Paul Andersen saying there have been no VAC setbacks to date. They are confident that they have a good package for the VACs including pricing, and ability to slot into existing HCPCS reimbursement codes.
The new metrics in the bottom half of the chart are shown for the first time, and the 70% repeat customer metric is encouraging at such an early stage.
On VACS Paul pointed out that some meet only quarterly, some twice a year, and some annually, so it will be a rolling process that continues over the next couple of years, progressively adding new accounts every month.
(A comment from me at this point: I think this is something that investors have to understand with medical devices. Pharmaceuticals typically ramp faster than medical devices after FDA approval because they rely on centralised reimbursement, simple prescribing behaviour, and scalable promotion, whereas medical devices face fragmented hospital procurement, procedural inertia, surgeon decisions and retraining, and local reimbursement friction. Drugs often show a 12–36-month steep uptake curve; devices more often follow a 36–72-month grind, unless they are capital-free consumables with strong reimbursement and minimal workflow disruption.)
Overall, I am pleased that $OCC have given this very granular level of detail as to the baseline towards the end of CY2025, as it provides a clear reference against which investors can track progress through the next year or two.
2. Understanding US potetial in the context of progress in ANZ
Although $OCC have made the comparison between the ANZ experience and the US market before, I think today they communicated the key ideas more succinctly and clearly. Consider the next slide:
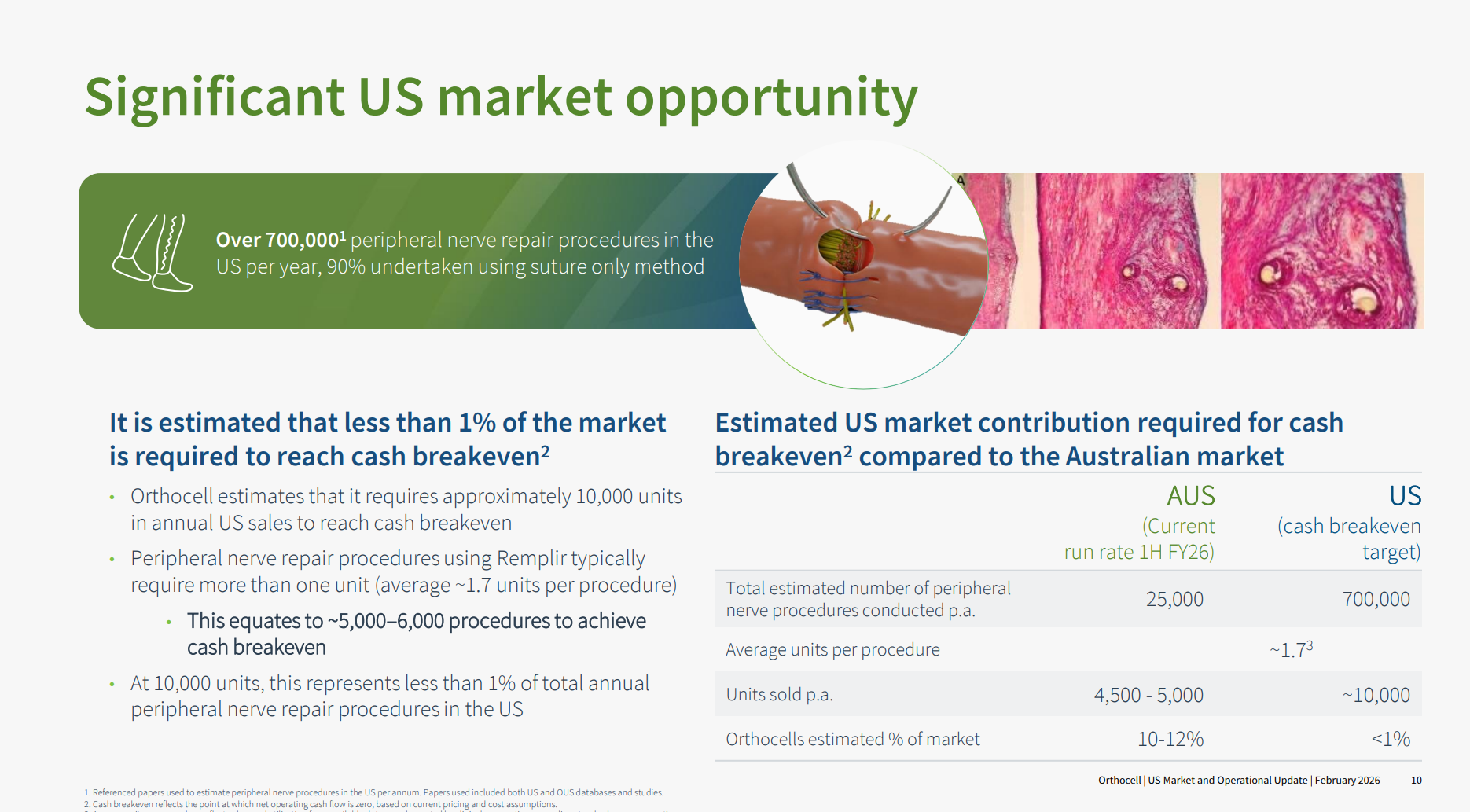
Implants to aid peripheral nerve repair make up only about 10% of the 700,000 annual procedures in the US. Here Axogen has the lion's share of the implant market (and it is worth following the progress they've made over the last 5+ years).
$OCC believe that they have evidence showing that Remplir is a superior product in the clincal outcomes (more flexible), but that in any event, they are focused on addressing the 90% of the addressible market using suturing as the established standard of care. Importantly, they argue that getting to cashflow breakeven requires capturing on 1% of the US market.
Drumroll .... what they've achieve in 3-4 years in ANZ: they estimate they have already succeeded in capturing 10-12% of that market.
On the call there was some discussion of the basis of their confidence that the clinical environment is sufficiently similar between the US and Australia, that they are confident they will also be successful in the US.
Both in ANZ and the US they are using distributors. In fact, today they rolled out the details of their hybrid US sales strategy, with a team of 7 in place in the US, to support dsitrubutors and hospital approvals. I think that makes a lot of sense, even though it will continue to drive SG&A growth in 2026.
From my unpicking of historical disclosures, ANZ Remplir revenue tracked $0.457m (FY23) and $1.04m (FY24) and thereafter it gets harder to unpick things. However, I believe that sets the stage for how we might assess US performance. With $0.09m sales in December as the first US revenue data point, what is really interesting to me will be the results for the March quarter and June quarter, as these will show how VAC approvals lead to accounts lead to sales, and then to repeat sales.
So, I consider 2025 as basically priming the funnel, with 2026 as the first full year where we will see revenue delivered from the US.
Quarterly US Remplir revenue numbers (which surely they will disclose?) will start to paint the picture of the trajectory that might be possible in FY27, and from that it will start to become possible to project when the 1% US market share might be achieved, and where Remplir can get to in that market 5-6 years down the track.
And that's the reason why I am totally ignoring the share price. $OCC has to be given the time to deliver in the US.
Yes, breakeven might still be some way down the track (FY28 or later) given that cash burn is around $6m per quarter or, let's say, $25m in round numbers annually. And it will be important to see how cash costs continue to grow in Q1 - for example, we might not have yet see the full quarters impact of the US team of 7.
So, it is early day to be investing in $OCC. My reason for getting in to early, is that there is the prospect of rapidly accelerating US revenue. I'm still not convinced we are going to see that, however, the progress presented today looks good and there are no negative flags from my perspective.
3. Capacity Expansion
$OCC flagged that in FY27 they will spend $5 to $5.5m to expand their WA manufacturing facility from 80,000 - 100,000 up to a first phase expansion of 300,000. (Note: that should support sales of some A$200+ million!)
Importantly, the investments will involve increased automation and the potential to run 24 hours operations. It is clear from the photos that the current operation is rather labour intensive, so as production expands this capital-light process should support strong gross margin expansion over the long term.
4. Clinical Developments
Paul Anderson promised an update on the use of Remplir in nerve-sparing in prostatectomies by way of a per-reviewed publication in Q1 2026. So one to keep an eye on.
They are inaddition collecting real world follow-up studies on a 59 paitent cohort, which will continue to add evidence to support their market growth.
Overall Takeaways
I remain positive about the potential for $OCC to succeed oin the medium to longer term. Progress to date in the US hasn't yet met my criteria to increase my initial now 3.5% RL ASX position. (Why: I don't have enough market data to project low, mid and high scenarios for US revenue growth over the next 5-6 years.)
More generally, I think the market will also continue to adopt a "show me the money" view for the US.
There could be other catalysts through the year such as futher 1) clincal updates, 2) a strong positive result from the prostate trial, and 3) approvals in the EU/UK. However, these are all "jam tomorrow" newsflow, and really, I think the success or failure of $OCC as a 3-5 year investment proposition is riding heavily on US sales momentum that develops in CY2026.
It is still early days, and I think there will be ample time to get onboard this one further down the track.
Disc: Held (RL 3.5%)

