Island is a drug re-purposing company with two core assets with short to medium term approval and commercialisation prospects:
- Galidesivir: A strategic antiviral targeting counter bioterroist agents stockpile demands for treatments for Marburg (lead), Ebola and Sudan with possible commercial incomes and PRV asset as early as mid CY27. One pivotal animal study away from NDA, cost about US$4m.
- ISLA-101: Phase 2a treatment for Dangue, seeking partners and planning for Phase 2b. Company IPO’ed in April 2021 with a view to commercialise this asset which was ready for Phase 2a with Phase 1 safety passed. Possible PRV on approval.
I am ignoring ISLA-101 and focusing value on Galidesivir because the efficacy of ISLA-101 is still questionable (mid Phase 2), while Galidesivir efficacy looks compelling (all animals die without it and live with it) and ISLA-101 is 3+ years and US$50m+ in clinical costs away from NDA, while Galidesivir is likely to have an NDA in under 18 months and at a cost of ~US$5m.
Thesis Summary: ILA effectively has a late Phase 3 asset in Galidesivir with a clear path to commercialisation with compelling economics offering value many multiples of the current market cap, plus a likely PRV which alone underpins the current market cap.
Galidesivir Value
The company has just herd back from the FDA and been provided very clear guidance on the path to approval for Galidesivir. They confirmed that they are eligible for a PRV and the only clinical tests to go before submission is an animal trial using the animal rule on account of the ethical and practical issues of testing drugs to treat Marburg (and later Ebola and Sudan). The expected timeline is below:

The company expects the animal trial to cost around US$4m (A$6m) and US$1.5m in milestone payments they will need to made pre-revenue. They have just raise A$9m and had ~A$6m cash at the end of December, so they have the cash to push Galidesivir to NDA in 18 months if they keep any other clinical work to a minimum. However, there may be costs to set up manufacturing (unless they contract manufacture), so another capital raise is likely in the absence of NDF (Non-Dilutive Funding) in the form of grants – which they may well get. The US government has already thrown US$70m at the compound to have it fail in treating Covid, so what’s another US$5-10m?
So let’s assume another 10% dilution for the possibility of a cap raise and in 18 months they have an NDA plus are awarded a PRV which they sell at the end of CY28 for US$150m (could get US$200m) and get US$200m a year from FY28 in orders for strategic reserve stocks for 5 years only.
25% of the PRV goes back to BioCryst Pharmaceuticals who as part of the purchase deal and Royalties of 5-10% (lets say 10% to be conservative):

So, US$112.5m for the PRV and US$180m in revenue for 5 years, starting CY28. They require next to no marketing or sales, so operating costs are minimal and there is no Gross To Net deductions due to insurance or distributor costs, they are selling directly on contract to the US Government and possibly other governments. COS & direct Operating costs are going to be minimal, but let’s assume 30% of revenue but also assume they have indirect and unrelated costs (eg ISLA-101 development) of US$20m a year and increasing at 10%.
Assume the company runs to CY32 then closes (ie ISLA-101 has been a black hold), below are the numbers you get for a 15% discount rate (IV=A$0.96 a share), the current price of $0.40 indicates a ~40% annualised return. If the company keeps all the money and just pays out at the end of CY32 then the PV is $0.69 a share.
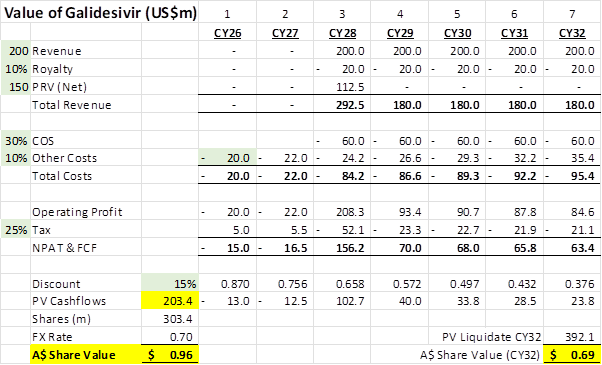
Note that the PRV is about 1/3 of the value in this scenario. The sales of US$200m a year is a suggestion from the company which seems reasonable in the context that the US government has provided ~US$600m in support for development of Marburg, which Galidesivir treats. Also, the US$200m is for a Marburg treatment only, the addition of Ebola and Sudan are expected to add US$100m each. Why Marburg I hear you say, well it’s the only Category A biothreat that has no treatment presently available in the US Strategic National Stockpile. Covered in the December update:
Island Pharmaceuticals (ASX:ILA) Webinar: FDA Response De-Risks Galidesivir Program Pathway
The low cost and speed of the animal pathway Vs normal Phase 3 and the direct to government contract sale process are significant differentiators to what a normal Biotech encounters when commercialising. There is a ready customer (US Govt) which will allow peak sales from day 1 and you save at least 2 years in the clinic, plus in this case they will get a priority review.
Conclusion
There is always a non-zero risk Galidesivir will fail to be approved, it seems unlikely but it has to be recognised it’s potentially a binary outcome if we ignore any value of ISLA-101, which I have. With this in mind I see it as a risk worth taking for the reward on offer and in general see this as an asymmetric investment base on probabilities and value. Also, the clinical risk is offset by a highly probable and clear commercial outcome on success.
Disc: I own RL having purchased this week a small position I intend on building.




