4 x trial sites In the coming months

Racura Oncology Principal Scientist, Dr. Rodney Cusack commented: "With governance approval now secured, we are able to move forward with the clinical trial of RC220 in patients with EGFR-mutated NSCLC. This approval confirms that the necessary site infrastructure, resources and oversight are in place, allowing us to progress safely and efficiently.
We remain focused on evaluating the potential of RC220 as an RNA and DNA G4 binder to target key pathways of osimertinib resistance. This important milestone brings us one step closer to making a meaningful impact for patients." Teams from Racura Oncology,
Monash Health, and Contract Research Organisation (CRO) Beyond Drug Development are scheduled to meet at Monash Medical Centre (Clayton, Victoria) for site initiation and training on 23 March 2026 under the supervision of Principal Investigator Dr Surein Arulananda.
Completion of this site training and subsequent site activation will enable the first patients to be recruited into the trial.
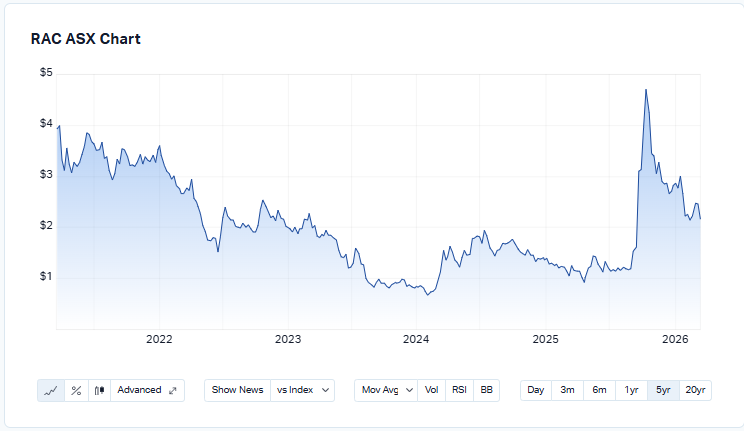
Return (inc div) 1yr: 110.68% 3yr: 1.75% pa 5yr: -11.51% pa