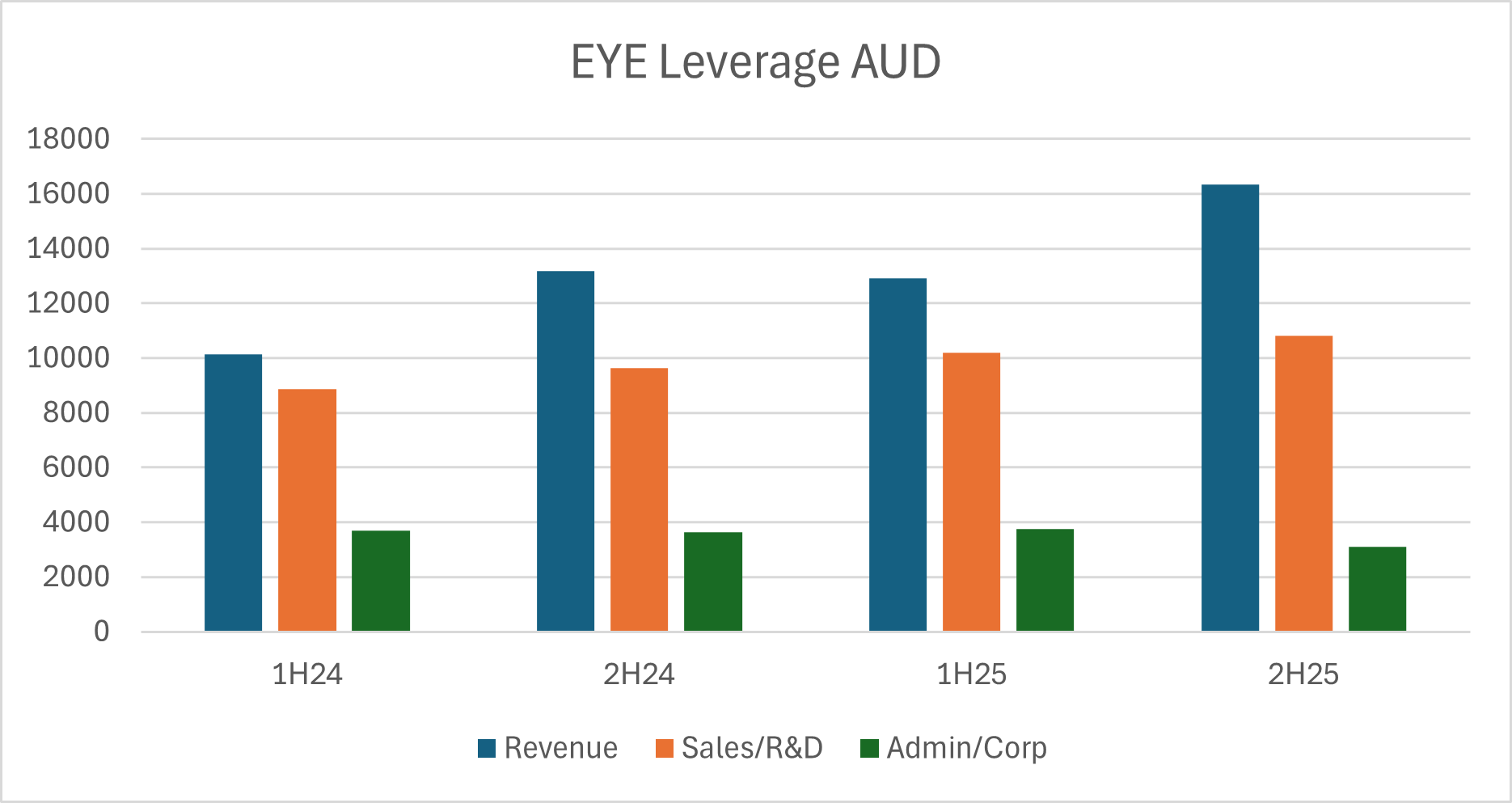Pinned straw:
While I am still getting my eye in on ophthalmology and the treatments for glaucoma, I did take a moment today to review the publication of the latest iTrack Register Study, announced this afternoon by $EYE, in response to the rallying cry from our fearless leader @Strawman, but also as a remedy for my own eyes for all the reading I have been doing about SaaS and AI!
Because I am somewhat of a science geek, I also has a read of the peer-reviewed journal article published in the American Journal of Ophthalmology.
And I got some help from my BA to boil it all down into the following, so that it is digestible.
Disease Background
Before getting into this, it’s worth recalling that glaucoma is an eye condition in which the normal system for draining fluid from the eye starts to fail. As fluid builds up, pressure inside the eye increases. Over time, this pressure can damage the optic nerve, leading to gradual vision loss and, if untreated, blindness. The risk of glaucoma increases with age, and it affects an estimated 76–80 million people worldwide. Treatment usually aims to lower eye pressure, most commonly with eye-drop medications, or with surgery that improves fluid drainage—by cutting tissue, inserting tiny stents, or clearing the eye’s natural drainage channels (a procedure called canaloplasty).
OK, now we can move on.
Summary of the iTrack Registry study
What was studied?
Doctors tracked real-world patients with glaucoma who underwent ab-interno canaloplasty using iTrack (a procedure that dilates the eye’s natural drainage canal). Most patients had the procedure at the same time as cataract surgery. The study reports around 800 patients and 1000 eyes.
What was the goal?
To see whether iTrack:
- Lowers eye pressure (IOP)
- Reduces the need for glaucoma eye drops
- Does so safely, in routine clinical practice
What did the study find?
1. Eye pressure went down, modestly but meaningfully
iTrack doesn’t dramatically reduce pressure, but it reliably nudges it lower, especially when starting pressure is higher.
- Average eye pressure fell by about 3 mmHg
- This reduction was durable, lasting close to 2 years
- Patients with higher starting pressure saw bigger reductions
2. Many patients needed fewer eye drops
For many patients, the main benefit was less reliance on daily drops, not extreme pressure lowering.
- Patients used about one fewer medication on average
- Around 40% of patients were drop-free at follow-up
3. Safety was very good
This is a low-risk procedure compared with more aggressive glaucoma surgeries.
- Serious complications were rare
- No vision-threatening events linked to the device
- Only a small minority later needed additional glaucoma procedures
What makes this study different from many MIGS trials?
- No medication washout. (Note: a “medication washout” is common in clincal trials. It involves a period before surgery when patients stop their eye drop medication, so that the “natural” state of the eye forms the measurement baseline, against which the impact of the surgery is measured. Because of this, “real world” studies that don’t have a washout, record lower pressure reductions post-surgery. This is on of several factors that makes it hard to draw comparisons with data from other trials for other treatments.)
- Patients stayed on their usual drops, so results reflect real-world practice, not idealised trial conditions.
- No cutting of eye tissue
- The procedure dilates the drainage system but does not remove or cut tissue.
- Broad patient mix
- The registry includes different surgeons, centres, and patient types — not a narrow trial population.
What the Study Indicates about the Comparison with OMNI – its canaloplasty competitor (Manufactured by Sight Sciences, where as flagged by @Wini, iTrack is gaining market share in theUS without, IMHO, pushing very hard.)
OMNI provides slightly greater pressure reduction, particularly in higher-pressure eyes, while iTrack delivers comparable final pressures and meaningful medication reduction with a simpler, tissue-sparing procedure and excellent real-world safety.
Key points
- OMNI lowers eye pressure more on paper (≈7–8 mmHg in trials), but this is largely due to medication washout before surgery.
- iTrack lowers pressure by ≈3 mmHg in real-world practice, with final pressures similar to OMNI (≈14–16 mmHg).
- When washout effects are adjusted for, OMNI’s true advantage is likely only ~1–2 mmHg.
- OMNI achieves higher drop-free rates in trials (≈70–80%) versus ≈40–45% for iTrack, though trial protocols actively withdraw medications.
- iTrack is tissue-sparing and lower intensity, while OMNI adds trabeculotomy, which explains its modestly stronger pressure lowering.
What the study is not claiming
- It does not claim iTrack is the strongest pressure-lowering glaucoma surgery
- It does not replace trabeculectomy or tube surgery
- It is not designed to hit very low target pressures
Key Takeaway
In everyday clinical use, iTrack safely delivers modest but durable eye-pressure reduction and significantly reduces the need for glaucoma medications, making it well suited for mild–moderate glaucoma and medication-burden reduction.
When we try to compare the performance of iTrack with OMNI, it starts to flesh out the basis of competition. Simplicity and safety appear key features. It is likely that cost is also a factor, although that is more opaque (and not a finding of this study but covered in other posts).
And finally, $EYE have a laser focus at the momen, on their highest value market segment, and are achieving a strong return on sales and marketing investments. (No doubt the presses are already running on incorporating this latest update in the marketing materials.)
$EYE need to keep updating the iTrack register. Some of the other MIGS treatments how have history out to 6 years!
====================================================================================================================
AND FINALLY - more insight on the iTrack Register's "Independence"
Several days ago I posted about the iTrack Register and its claims to be "Indepdent". The good news about scietific journals is that they are very switched on to conflicts of interest, and require quite granular disclosures. So, here's what we've learned further about the iTrack Register and the studies that come from using the data, courtesy of the Americal Journal of Ophthalmology:
I quote without editing:
"ACKNOWLEDGMENTS / DISCLOSURES
a) Funding/Support. No funding was gathered or allocated for this study. Surgeons were offered a reimbursement from Nova Eye for data entry of each follow-up visit. No other compensation was provided.
b) Financial Disclosures.
The authors have no financial interest in the products mentioned in this study. Drs. NK, DL, KB, SP, KM, IIA, KM are consultants to Nova Eye Medical. Drs KB and NK are the founders of the IGSR.
c) Other Acknowledgements.
Writing and editorial support were provided by GP Communications and funded by Nova Eye Medical. The funder bore the cost associated with the hosting of the iTrack™ database by the IGSR, but was not involved with the data collection, analysis, or interpretation, and did not influence the results of the study.
"The authors declare the following financial interests/personal relationships which may be considered as potential competing interests:
Iqbal Ike K. Ahmed reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ahmad A. Aref reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Keith Barton reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. Keith Barton reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Jason Cheng reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. Jason Cheng reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper
Paul Harasymowycz reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. Paul Harasymowycz reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Nathan Kerr reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. Nathan Kerr reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
David Lubeck reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. David Lubeck reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Karl Mercieca reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. Karl Mercieca reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Shamil Patel reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. Shamil Patel reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Nir Shoham-Hazon reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. Nir Shoham-Hazon reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Harmanjit Singh reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Justin Spaulding reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. Justin Spaulding reports a relationship with Nova Eye Medical that includes: consulting or advisory and non-financial support. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Samuel Thomsen reports financial support, administrative support, and writing assistance were provided by Nova Eye Medical. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper."
Disc: Not held, but watching
@Wini first up ophthalmics is not an area I've looked at in any detail before (get it?... but true.) So have that caveat in mind as you read what follows.
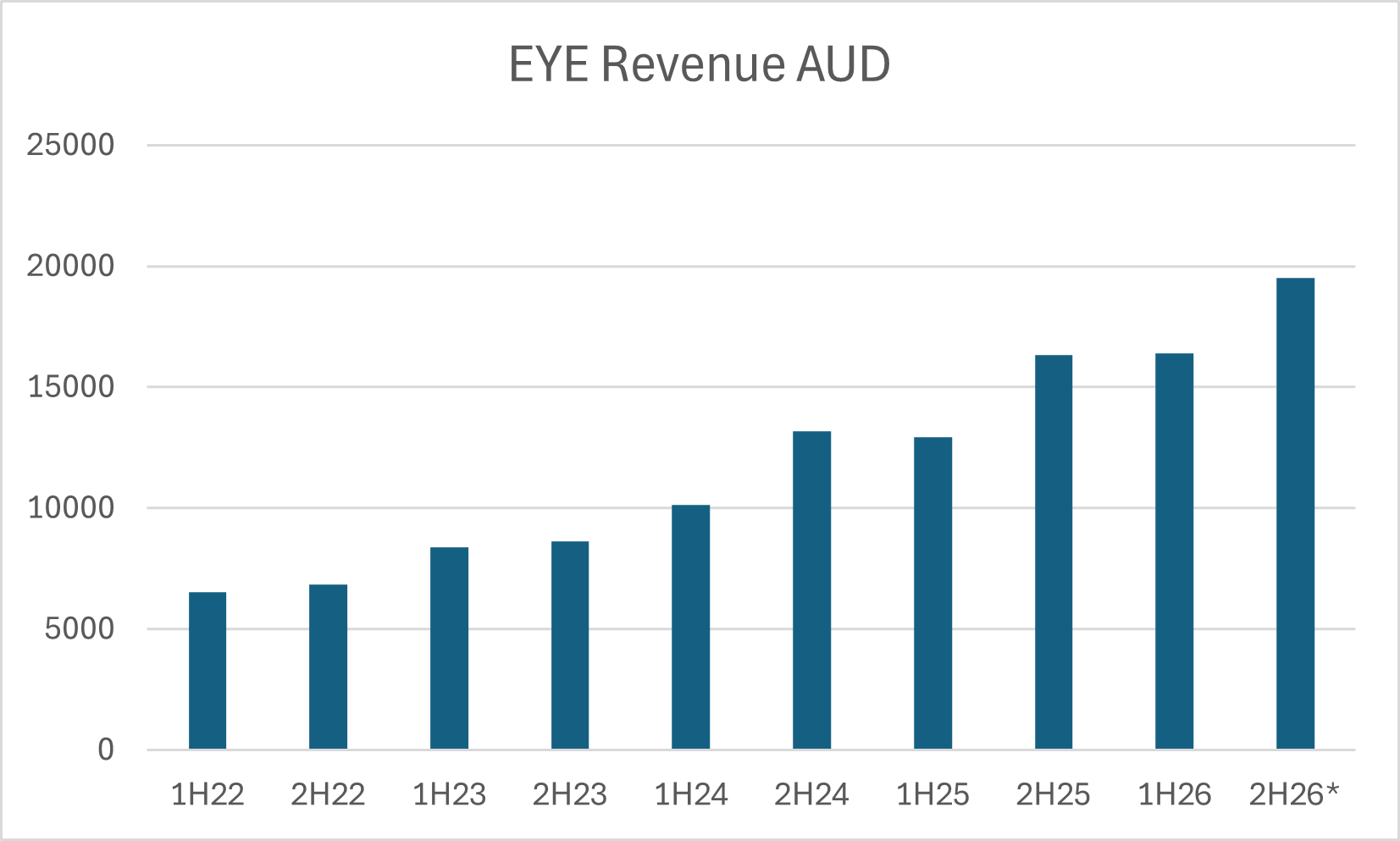
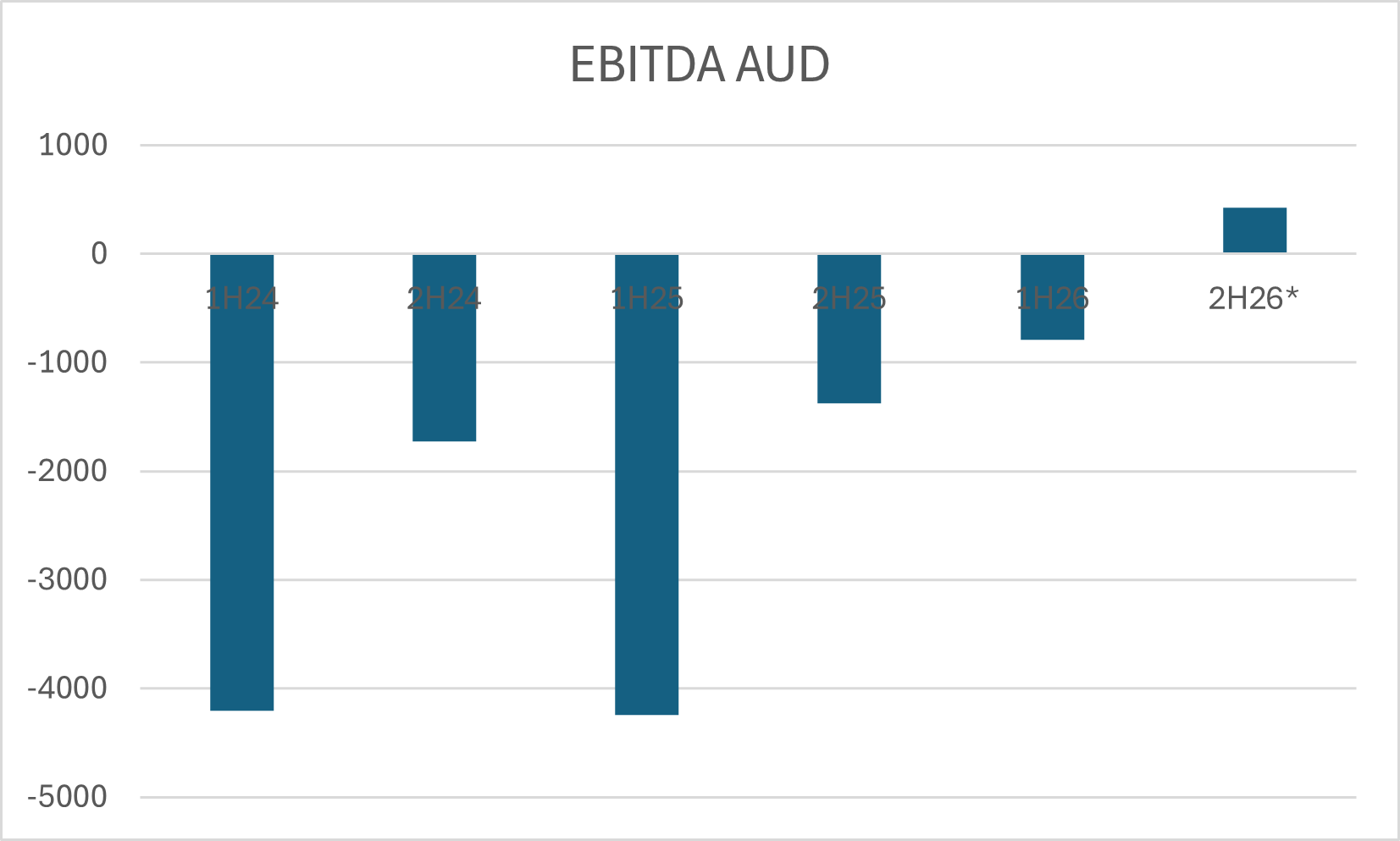
$EYE looks like a steady developer, moving to profitability and, with valuation today around 1.5xRevenue 'ish, if they can continue to scale revenues at 20-25% annually, while maintaining good cost control, then there could be a nice re-rating of the SP over the next year or two. So, just goofing around with some numbers, if they continue to grow revenue at 25% for a couple of years, they'll get to $45m sales in FY27, and if costs are well controlled that could get them to an NPAT of $6-8m. If they then were rated at a P/E of 25, which would be undemanding given the revenue growth, market cap would be $150m - $200m, up from around $55m today.
So it is a classic profitability inflection re-rating opportunity. And a good time to look at it.
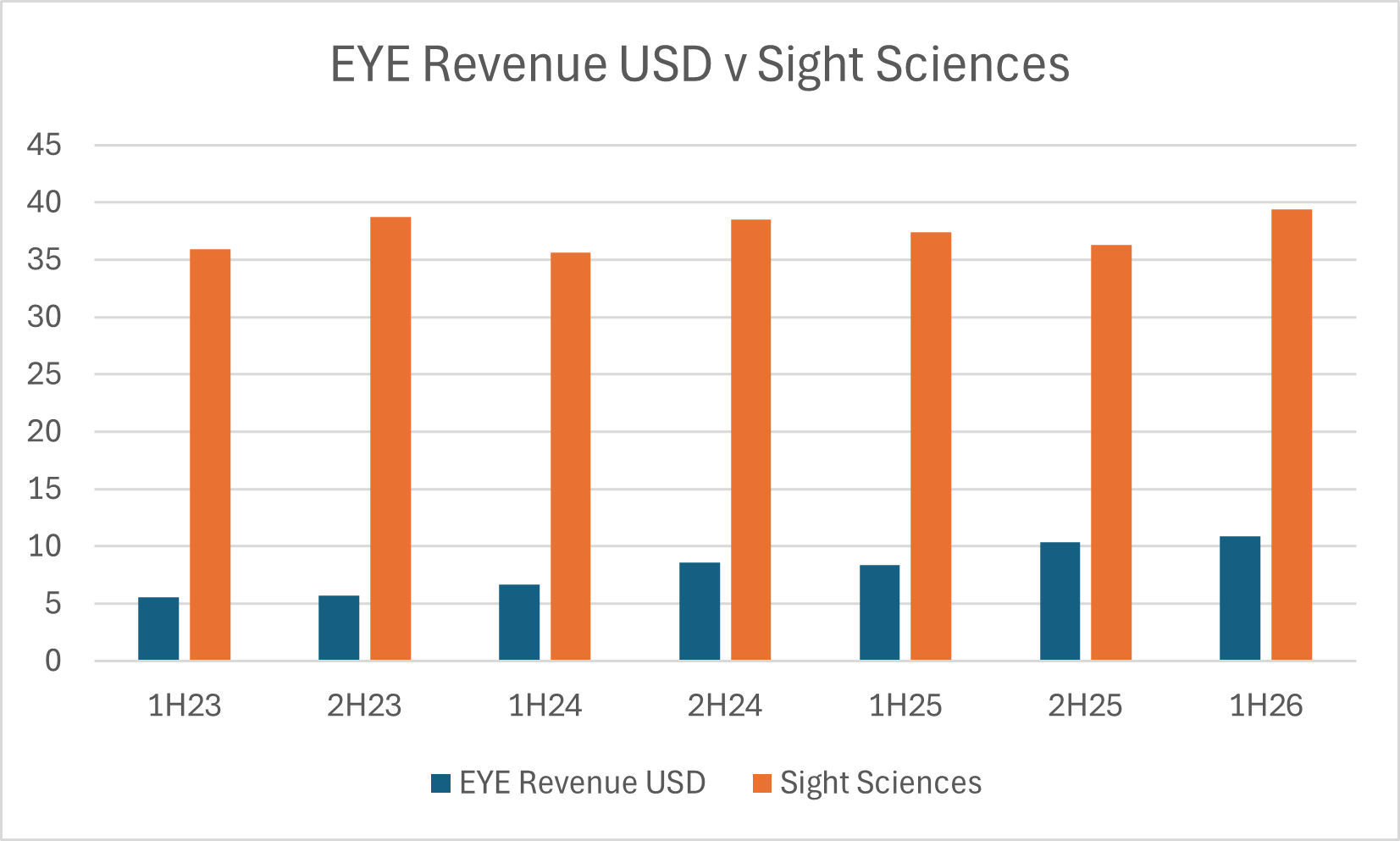
One thing I'd want to understand better, is why isn't Sight Science's revenue growing?
From a quick look at the market, according to Grandview Research, the global MIGS market appears to be growing at 5.4% annually over 2023-2030 with the US and RoW apparently at the same rate. As ever, per procedure revenues will be most attractive in the US.
From a quick analysis of 2021 and 2022 US Medicare data, canalplasty procedures rose by 47.4%, whereas angle-based stenting fell by 28.6%. However, stenting remains much larger share of the market than canalplasty. While these are numbers from only one year in the US, they do provide some evidence of market headroom for $EYE, while calling into question why Sight Sciences is so flat. It is possible to do a deeper dive into this area, but this is just what I found with 5 minutes of work.
So, this is one to add to my research list, and I will report back with what I find, although probably in a few weeks/months.
Thanks for flagging it!
Disc: Not held