Pinned straw:
$RMD posted their 2Q FY26 results this morning. I'll share their headlines, and then summarise a few of my takeaways from the investor call just finished.
TLDR: a solid result with a few pluses and minuses. %GM continues to do heavy lifting. (+310bps PCP; +30bps QoQ) Overall GAAP diluted EPS broadly inline with consensus. NPATgrowth of +14% / +15% for diluted EPS due to $175m of buybacks in the Q.
All numbers are US$, except for my valuation.
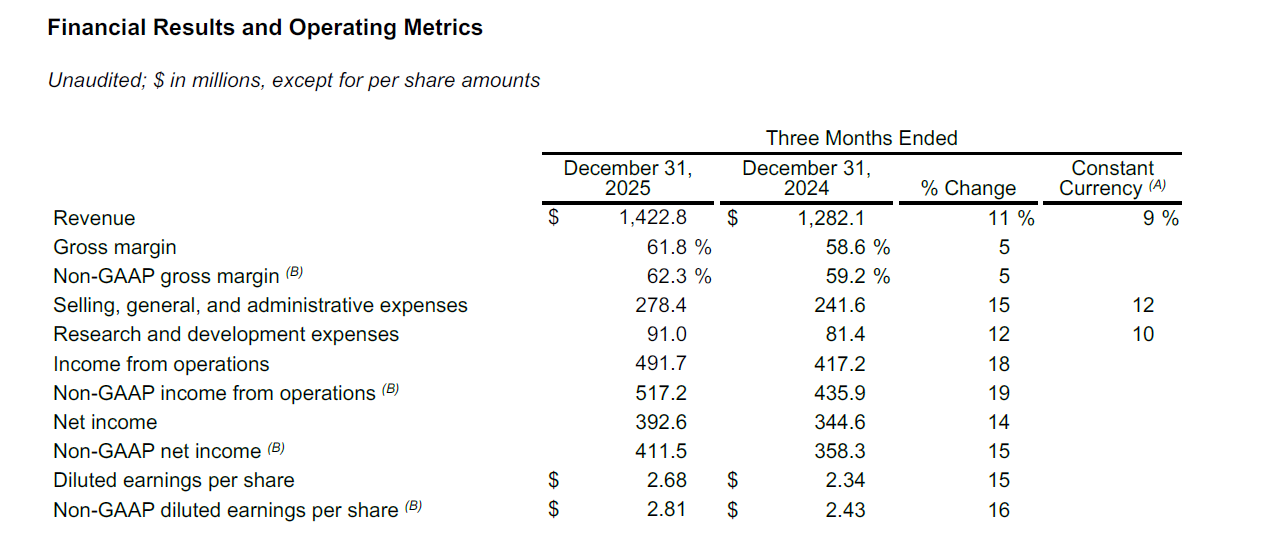
Their Headlines
• Revenue increased by 11% to $1.4 billion; up 9% on a constant currency basis
• Gross margin up 320 bps to 61.8%; non-GAAP gross margin up 310 bps to 62.3%
• Income from operations increased 18%; non-GAAP income from operations up 19%
• Diluted earnings per share of $2.68; non-GAAP diluted earnings per share of $2.81
• Operating cash flow of $340 million
Observations from the Investor Call
1. Revenue (all % to PCP)
Devices growth in US +8% strong, weaker in RoW at =5% (CC) explained due to cycling a strong PCP (+9%) and lumpy effects (e.g. fleet management in Japan).
Mask +16% in US a strong result, with +8% cc RoW also weaker. Overall mask strength OF +14%(CC).
Masks starting to see benefit of new fabric masks, with reported positive user feedback.
2. Gross Margin
%GM of 61.6% up 310bps (PCP) and 30bps (QoQ).
Mick spoke of continuing focus on %GM expansion, with goal of sustainably driving this up, and with a set of identified improvements in hand.
90% of US customers now able to receive orders from US factories within 2 day.
3. Expenses
SG&A was a drag on result up +12% (CC) actually higher than revenue growth. Continuing marketing campaigns cited as one ongoing driver - as $RMD are active in experimenting with new ways to reach customers and promote awareness of sleep health. A $6m exceptional expense saw the conclusion of costs of restructuing.
These two items together pulled Operating Income growth down slightly to +17%.
4. Balance Sheet
Cash and cash equivalents up to $1.417bn from $1.383bn in prior quarter
Net cash is $753m, up from $715m in the previous quarter and significantly up from net debt of $151m in the PCP.
So despite $88m in dividends paid and $175 million share repurchases in the quarter, the balance sheet has strengthened significantly over the year, and incrementally over the quarter. Expected buyback of $600m in FY26.
Other Notes including from Q&A
3-yr result on increased resupply rate of +6% for patients with both a CPAP and GLP-1 script announced a few weeks ago at the JP Morgan Conference was reiterated. Mick believes the GLP-1 headwind is proving to be a tailwind.
He was asked whether int he dataset, this remained true for mild of moderate AHI scores, which can be significantly reduced by GLP1, and Mick said you still need CPAP for the symptom relief and improved sleep, and promised to publish "case studies" in future. But he did not commit to segmenting the real world evidence.
$RMD have FDA approval for their sleep concierge MyAir. This uses AI to give personalised recommendations to users on how to optimise the machine settings. Mick said the $RMD devices have settings that the large majority of users have not used for over decades. He believes personalised settings will improve patient experience and adherence, keeping more patients on devices for longer.
Mick discussed new publications showing benefits of CPAP therapy on brain health. For example, adherence to CPAP may reduce dementia risk by 30-40%, based on evidence of impact on reducing key Alzheimer biomarkers.
Mick said this is adding to the growing body of evidence that CPAP drives sleep health, cardiovascular health and brain health.
On tariffs, $RMD products have continued to retain exemptions from international tarrifs, and he repeated the news from JP Morgan Cont that $RMD products will be exempt from public health competitive tendering in the US.
No change on the messages regarding the return of Phillips. Mick quoted Philips management as saying that they still don't know when they can return to the US. And in the 100+ international markets where they have returned, Mick said $RMD has seen no change in its market share, and that Phillips is fighting to recover #4, #3 and #2 spots. Key message: we welcome competion but see the ecosystem of devices, masks and software are strong.
Mick believes consumer health will continue to drive patient funnel. Expects more wearables to add OSA detection FDA approval (adding to Apple and Samsung).
My Overall Assessment
A solid, relatively uneventful result.
Things to watch in future:
- SG&A growth - will it normalise?
- RoW devices and masks - does it tick back up?
- GLP-1: what granularity is disclosed for the mild/moderate cohort?
Valuation
Unchanged at A$42.00 (A$37.80 - 46.70) - next update in October.
I'm a "BUY" on RMD, but as I have a 10% RL position, I am my target weight so am not adding today, but will consider some minor additions on any SP weakness, e.g., below $36.
Bushmanpat
@Solvetheriddle @mikebrisy and @Chagsy
How do you think Apnimed's oral pill treatment for sleep apnea will impact ResMed. It appears to be getting good results and a pill before bed is much easier than a CPAP mask.
mikebrisy
@Bushmanpat in trials it is showing a meaningful AHI reduction in a significant proportion of participants … median %AHI reduction between 45-50% from memory. Overall, it is well tolerated and while there are a range of side effects, the safety profile looks OK.
It looks like it is going to offer an alternative for those who can’t tolerate or refuse CPAP.
And like GLP-1, it may prove an alternative for some of those with mild to moderate OSA.
Mick Farrell, I recall, has said that he believes that combination CPAP and GLP-1 will be the gold standard.
But who can say, as we’ve only seen top line results from the Apnimed Phase 3, and we need to wait the complete analysis.
Overall, based on what I know at the moment, I don’t think it materially, additionally reduces the market for $RMD than the impact the GLP-1s will already have. And as ever, the availability of treatments is going to put more patients in the funnel through general awareness, and for some of those patients, CPAP or combination therapy will be best.
I don’t think anyone knows at the moment. So it is just one to watch.
The other question is do more effective treatments emerge over time.
Bushmanpat
@mikebrisy and I guess if/when it does hit the market, the price point will it enter at will determine the impact on RMD.
mikebrisy
@Bushmanpat … well the comparison is that the cost of GLP-1 is 10-20x the ongoing cost of CPAP consumables, and the setup cost of CPAP is only a couple of months of GLP-1.
Of course, GLP-1s prices will come down over time.
So I imagine the two extremes anchor where the Apnimed product is priced.
Of course, it will also depend on the label indication. I.e. how broad or narrow.
Lot’s of unknowns at the moment.
Karmast
I also wonder whether Apnimed is an acquisition target for Resmed?
If the Apnimed results continue to be good and they aren't crazy with an asking price, a genuinely successful drug option under the Resmed parent company, makes for a mighty powerful global leader...
Mike is a smart guy and they would have to at least be discussing this. They have about $1.4 billion in cash and $660 million in debt, so there is some firepower there and they'd also get some support for a raising at a sensible price if necessary. I believe there are a number of other drugs in trials too, so getting going on the first proven one and making it the first choice with Dr's through their global distribution network is probably a savvy move.
Solvetheriddle
@Bushmanpat maybe that's a reason RMD appears good value. How do i class these risks? I'm in the wait-and-see how they progress. RMD, I believe, has a holding in apnimed, so they are watching as well. The framework here is you can buy a company with no hairs and pay a monstrous price, eg PME, buy one at a fair or good price with some concerns, but no evidence of impact its a maybe situation (that's where i like to sit), or buy a company thats is being eaten alive, but you get a low SP. i just think its too hard to assess at this stage and i want more evidence. BTW we will see this fear in droves with AI, IMO.
mikebrisy
@Solvetheriddle @Bushmanpat yes, Apnimed ... and the prospect of the emergence of a class of OSA-management pills ... is without doubt weighing on $RMD's valuation.
And, yes, $RMD does have a minority stake in Apnimed. Apnimed is private, and so details of the holding have never been disclosed.
Not only that, Carlos Nunez MD - the Chief Medical Officer at $RMD - is on the Scientific Advisrory Board at Apnimed.
It is very clear from numerous statements by Mick Farrell over recent years, that they are watching the progress at Apnimed very, very closely.
More recently, there has been a shift in the strategy focus and messaging of $RMD from treating OSA with CPAP, to being a leader in sleep health.
I have no doubt that if $RMD conclude that the Apnimed opportunity is attractive, then an acquisition would be considered. So I am with @Karmast there. And, in fact, as the cash pile grows it risks burning a hole in the pocket. Mick has made clear that they continue to actively consider acquisitions.
So, part of my consideration is that I believe Mick and his team are far more qualified to judge the risk of the OSA-pill to the CPAP business than I am. With Nunez close to being on the inside, they are well-positioned to moving at the right time.
As GLP-1's are showing, the availability of alternative and complementary treatments might not necessarily be a threat to $RMD's continued success.
Bushmanpat
@mikebrisy @Solvetheriddle @Karmast Very valuable responses. This is the second level thinking that the SM community is so good at. It looks more likely to me now that the worry is the opportunity, but RMD isn't a set and forget stock.
thunderhead
+1. It looks like as is typical, Mick and ResMed have positioned themselves well to "see around the corner" with regard to this evolving threat, and have the means to take it out if necessary.
Chagsy
Thanks as always @mikebrisy
I see the “lack of granularity” as an orange flag.
My concern is that with the advent of oral GLP-1 agents hitting the market, the barrier to patient uptake is likely reduced. Meaning the mild - mod overweight and by extension OSA and potential CPAP users may actually not take up CPAP. This group are perhaps less likely to be as motivated as the significantly obese and sever OSA sufferers
I know that weight is not perfectly correlated with OSA and that OSA occurs in the non obese, but this certainly something to watch.
Great margin results though!
mikebrisy
@Chagsy I agree.
My logic is: They have the data. If the result was favourable, they'd share it. If its not or unclear, they can say it has been disclosed in aggregate. Looking forward to seeking the "case studies" (is that language for cherry-picked?)
As oral GLP-1 hits let's see what happens. I'm happy to let the numbers do the talking, as last time I jumped at the GLP-1 phantom I forewent a 10-15% return!
One to watch while staying in the game IMO.
Chagsy
I've mulled this over all day @mikebrisy and figure, at a very high level, there are two opposing but not mutually exclusive concepts that need to be considered:
- when new innovations arrive, we generally over-estimate the near term impact, and under-estimate the long term impact. Witness the share price collapse of a few years ago - lucky me - I had been waiting for a decent entry point into Resmed for ages. However, I am slowly changing my mind on the impact of GLP-1 s on CPAP's future. I now think they will have a reasonable impact - predominantly in the less severe OSA/obesity category. I believe this will be slow and nearly imperceptible for a year or two, but then increase significantly as the data rolls in, the drugs become more accessible and the s/e profiles improve.
- countering this is the TAM. Currently, according to my Brain-for-hire (sorry I couldn't be bothered to double check all the numbers, so there are possible hallucinations in this), RESMED has maybe 10m active users of its devices. There are an estimated total OSA sufferers worldwide of ~ 1bn people! Of which nearly half a billion have severe OSA.
- say GLP-1s cut the TAM by 30%, (we're down to 660m) half of those won't ever be able to afford a sleep study or even a wearable to diagnose the condition, let alone the cost of purchasing a CPAP device (down to 330m) and half of those are not motivated to look after themselves (we are down to 165m). If Resmed keeps its market share of close to 50%, we still have over 80m people as a potential TAM. Of those, 40m will have severe OSA for which CPAP is life saving. Versus the current 10-15m.
These guesstimates are going to be wildly out, of course. But whichever way you chop the numbers down there is a still a good runway for growth.
This assumes a) Resmed's market share neither does up nor down. This assumes there is no new intervention to disrupt the marketplace.
So, yes, in summary, I think letting the numbers do the talking is a sensible option!
Best wishes
C
EDIT : half a billion, not half a million
mikebrisy
@Chagsy that’s pretty much where I’ve landed too.
And it explains why $RMD are spending so much on driving research and awareness (both with HCPs and the public) of the various benefits of sleep health. They are working on activating the maximum TAM.
Enjoy the skiing … cough … conference!
Solvetheriddle
@mikebrisy @Chagsy thanks for the comments im just catching up with RMD, with a pretty volatile US result season taking my time. The only thing i would add is that the US brokers appear to be doing channel checks to substantiate RMD claims of demand for GLP-1s helping RMD, and it appears to be consistent.
the US results are also consistent. my view its true until its not as shown in the numbers. might be helpful for AI hysteria as well
held --adding some recently
OxyBBear
RMD one of the few stocks in my portfolio down by a hefty percentage today of 4.5% after going ex dividend approx 6c.
Looks like the Phillips' Q4FY25 result may be the culprit based on competition returning as that is all I get from RBC Capital's take below.
RBC Capital: "We have analysed Philips’ (covered by RBC analyst Natalia Webster) 4Q25 result (Dec YE) for potential implications for RMD and NAN which include: "In 4Q25, the Connected Care segment reported 7% comparable sales increase, driven by double-digit growth in Monitoring and mid-single-digit growth in Enterprise Informatics. "However, in this result very few comments were made about the Philips’ Sleep & Respiratory Care business and therefore we did not obtain any read-throughs for RMD.
"In 4Q25, the Diagnosis & Treatment segment reported a comparable sales increase of 4% with Precision Diagnosis’ flat and double-digit growth in Image-Guided Therapy. "Management noted strong hospital demand in North America but noted consolidation among larger health systems, rising costs and workforce shortages. We consider this commentary to be a negative read-through for NAN."
mikebrisy
@OxyBBear I'm not reading anything into today's SP movement on $RMD. It mirrors US overnight.
Basically, post the latest quarterly, $RMD advanced from $35.73 up to $38.61. There were some minor TP moves up from various analysts,
Today they've then gone Ex-Div as you've said, and CEO Mick has done one of his small sell-downs (1.1% of his holdings) - neither of which on their own explain a -4.7% movement, but when you see the pre-results price, then a close today of $36.79 is neither here nor there.
For interest, below I've copied what Mick said about Phillips at last week's conference call. (Of course, it is Micks view and spin!) Important to remember that $RMD has several other competitors, for machines and masks, including the ASX's own $FPH. In several markets Phillips is having to claw its way back from number 4. So, one to keep an eye on, but not a biggie at the moment IMO.
----------------------------------
Extract from Earnings Transcript: Source S&P Capital IQ
David Bailey Morgan Stanley
One of the most frequent questions we get is the potential return of Philips back into the U.S. device market. Just thoughts on any potential timing of that reentry to the extent you've got any thoughts there? Are there potential impacts? And where they have reentered your observations in relation to the competitive dynamics, that would be great.
Michael Farrell CEO & Chairman
Yes. Thanks for the question, David. And I'll just default to what that company's CEO said at a conference earlier this month and what their CFO said at a conference I was at the quarter before, which is they don't know. So if they don't know, then I don't know. But -- and frankly, I don't really look back to the #2, 3 or 4 competitor, which that company is in the various markets, 140 markets we compete in.
I look forward to say the last 5 questions, how do we deal with this new flow of patients in. I can say this, though, they're back in over 100 countries in Europe, Asia, Rest of World, and some they've been back for 12, 18, 24-plus months so we've lapped it multiple times in these quarterly calls. And I don't know if you looked into the Europe, Asia, Rest of the World device growth in the quarter, pretty solid. They're at 5%, like right in line with market, maybe a little demand gen that we're driving. We're holding share and competing well with that competitor. But more importantly, frankly, in some of those regions, other competitors that have taken that #2 spot that, that competitor you mentioned has to fight with to get back to #4, #3 and even #2 space.
So I welcome competition. I think competition is fantastic. I'm not fearful at all about that player that is going to be making products that I think go in Thailand now and shipping them to the U.S. and Europe. I look forward to that. I think we've got a better product. It's small, it's quiet, it's more comfortable, it's more connected and it's more intelligent.
And that ecosystem, what we developed with myAir and AirView and Brightree, that ecosystem is very hard to match and none of our competitors really can compete across the board on that. But no, I have no idea of the U.S. devices entry, but they've been in the U.S. for masks and accessories these last 20 quarters, and we've been competing very well against them and meeting and beating their growth and taking share when that's all they could sell in that market. I'm very happy to have them back Monday morning or next year, and it really won't affect us in terms of how we grow to the market. They'll have to fight to get that #3 or #2 position from another player that took it, and we welcome that competition. Thanks for the question, David.
Disc: Held