Pinned straw:
The recording on the interview with Tom Spurling is now up (see members page), and you can access the transcript here: Nova Eye Transcript.pdf
I guess my initial takeaway is that this is a fairly clean thesis as far as med-tech goes. Nova Eye is post-commercialization with strong sales and revenue traction, albeit off a very small base, in its key US market. They have achieved a 40% CAGR over the last six halves while holding only a tiny percentage of the addressable market, which is around 3.5%. Tom noted they would be doing $60 million in revenue at 7% market share, a level he thinks they can capture in the medium term. There are also significant opportunities in other geographies, such as China where they have received clearance recently.
With the business apparently still on track for EBITDA breakeven this current half and no major CAPEX or cost increases evident, we should see strong growth in profits in the coming periods as 70% gross margins drop pretty much straight to the bottom line.
It is also nice that this is a relatively low-cost, quick turnaround surgical procedure without a huge price tag for the patient. There are plenty of potential patients given the high prevalence of glaucoma.
That said, the cash balance does not give them a lot of wiggle room, representing about seven to eight months of runway at the last half-year run rate. However, a working capital facility and recently reaffirmed guidance probably mean they will not need to raise cash. Tom gave the impression that profitability and cash flows were a major focus, even if that meant pursuing slightly slower growth than they could otherwise achieve.
Lastly, as has been noted by others, including Tom, the value of an established sales team that's generating good momentum and with a growing network of references is worth noting.
I have scheduled a call for us to talk through this business on Wednesday the 18th of February at 6pm AEDT. Keep an eye on your inbox or just check out the Meetings page. I am really keen to hear what others think, especially those who can emphasise some of the risks.
mikebrisy
@Strawman looking forward to watching the video of the Strawman Meeting.
But just to share an insight from my own visit this morning to my ophthalmologist (quite a young guy who considers himself an early adopter of treatments).
He is aware of the benefits of canaloplasty over the other MIGS treatments (e.g. using stents).
We looked up the Australian treatment codes and, according to the letter, canaloplasty's are not currently reimbursed, because the current code only related to procedures that leave a device behind in the eye. (e.g. stents, although the wording is more general that that).
My specialist noted that the FDA seems generally much more willing to register novel devices on a lower standard of evidence, including end points that favour the novel device, rather than head-to-head comarisons against the standard of care. He felt that in Australia and EU, reimbursement decision tended to hold out for more comparable evidence with approved standards of case. So, more often the answer was "No" to reimbursement.
Doing these head-to-head comparisons are expensive for a small company like $EYE (like the CEO says - I've read ahead in the transcript! - is it better to put on a few reps to get better US coverage, or to invest in a registration in another jurisdiction.)
My Dr felt that the investment case for $EYE will likely expand if the product moves out of the high volume, glaucoma-specialist centres, and into the more general opthalmology centres that do the canaloplasty at the same time as a cataract procedure. That will be most profitable for the clinics.
Apparently there is an ongoing debate in Australia about the code or codes that should be provided for in the treatment of cataracts and glaucoma, and there are different views in the profession.
Overall, my specialist says that the simplicity and low risk of canaloplasty means that he believes it will continue to grow its market share over time.
-----
So, I think that's the essence of this morning's "deep research" ... now to watch the $EYE meeting over lunch.
Disc: Not held
Wini
Appreciate the boots on the ground (eyes in the chair?) research @mikebrisy
Metis
@mikebrisy this is the most oddest measuring contest I have ever participated in but "my opthalmologist said" Hahaha
Source of information: two biased views from n =1 in the glaucoma surgeon I anaesthetise for and the rep who works for the competitor Alcon that make the nickel and titanium stent "hydrus" Who I chatted to yesterday. I'm sorry I have not had a chance to listen to the strawman meeting yet.
I'll try to explain the why and the how first:
Glaucoma: Causes irreversible blindness, due to increased pressure in the eye due to decreased drainage of its fluid. Often people are unaware until late stages. 2022 in US around 4 million people and in Aus about 300,000. It is a disease that worsens with age.
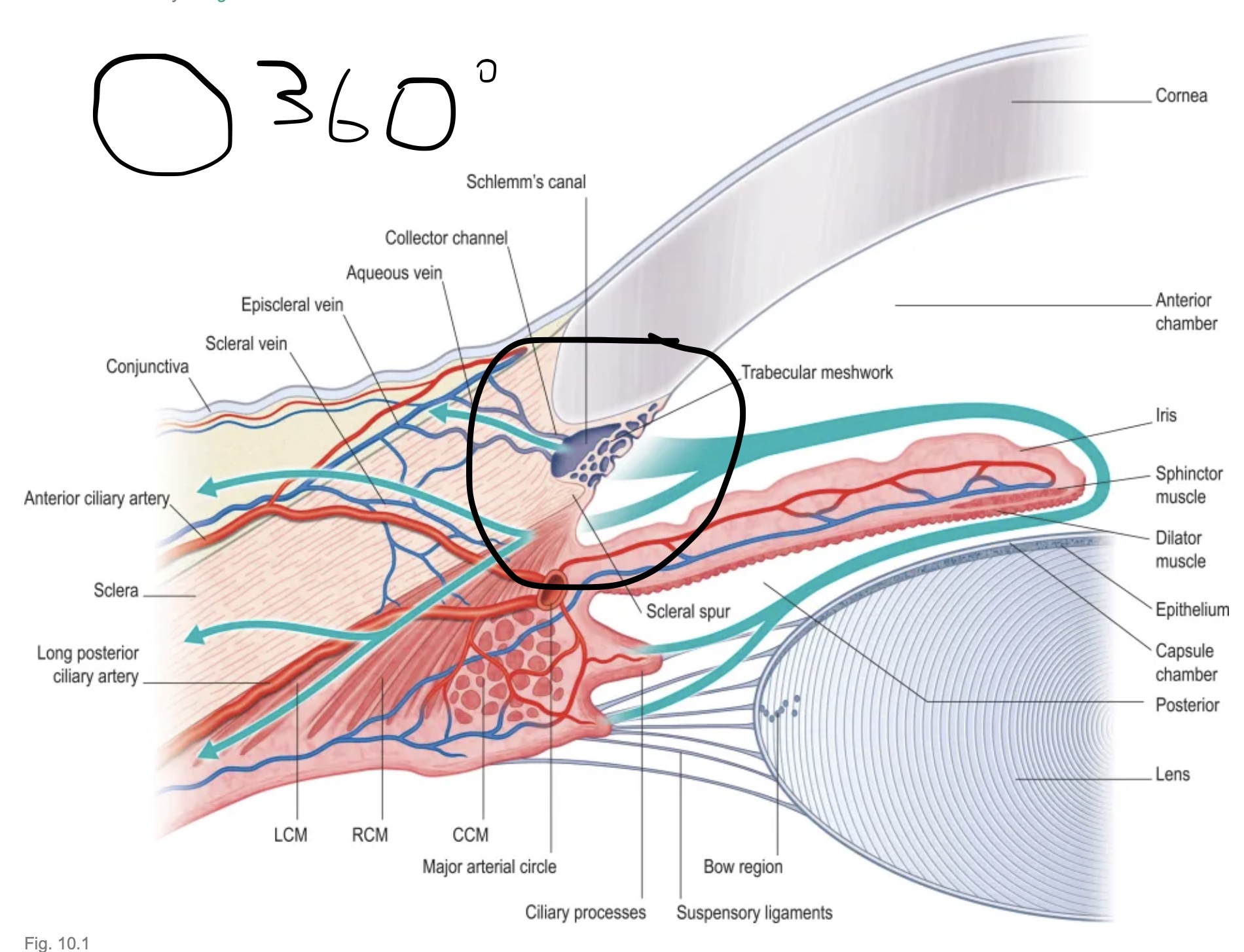
The treatment Deals with the flow of aqueous humor (fluid in eye). At the bottom of the post is the direction of flow with a picture. But for want of a better way to explain it, think of the main way for this fluid to be drained out of the eye is via a hula hoop (Called the Canal of Schlemm). The fluid enters on the inside of the hoop and on the outside it leaves via small and large canals.
- Now imagine that that hula hoop is squashed via either damage to the inside of the hula hoop (trabecular network) or issues with the hula hoop itself. There is decreased flow and more fluid kept in the eye and therefore more pressure which decreases blood flow to back of the eye.
The treatments try to help this problem of drainage:
I think it is important to clarify before going through the types of treatment that Australian ophthalmologists routinely do there fellowships in England and Canada so therefore the treatments often mirror those places. This is important as a surgeon learns one technique and often doesn't change it if it took him a year to learn that specialised technique. Hence one of the reasons for the slow pace of adoption of medical devices vs drugs.
Types of treatment:
- For Severe Glaucoma: Bleb treatment (invasive)
- Preseflo (not in the US)
- Trabeculectomy (1.5 hour long operation)
- Ahmed stent (Also 1.5 hour long operation)
- Molteno3. (NOVA EYE MEDICAL) difference with Ahmed stent is that it doesn't have a valve. The surgeon said "made by guy in new zealand not used anymore, Ahmed copied it and made it better."
The surgeon I work with uses the top three. Says no one HE knows uses Molteno3. (About 150-200 glaucoma surgeons in Aus). 1200 ophthalmologists.
Now for other treatment we have MIGS (minimally invasive glaucoma surgery): I see these as they put them in when they do a cataract. Usually 1 hydrus stent goes in each time I work with him. Often 10-14 cataracts on the list, they can be done as stand alone but often the cataract is done at the same time as it helps the glaucoma too.
- Hydrus: $1264 AUS dollars (HORIZON trial has beneficial data to 5 years). Nickel/titanium stent that essentially punctures into the hula hoop and stents it open for 90 degrees. Allowing access to multiple exit points for fluid both big and small canals.
- Istent: variable outcome (according to competitors rep) $1800 AUS. Has three puncture points into that metaphorical hula hoop however who knows what point it lines up with and may not line up with a large channel, just a small one. As any plumber will tell you flow is proportional to the radius to the power of 4. (poiseuille's law) So your three puncture points may only line up with small channels and have decreased drainage or visa versa.
- Omni - not a device, essentially it rips the trabeculectomy (inside of the hula hoop) over 180 degrees. Reimbursed for only twice a year.
- Itrack (NOVA EYE) aims at increasing the drainage 360 degrees around canal of schlemm. But doesn't leave anything in apart from a viscous elastic. Not reimbursed yet is my understanding. The surgeon I work with can only think of 1-2 surgeons who may use it. Doesn't plan to use it anytime soon. Just came back from the Australian/new zealand glaucoma conference and doesn't remember any presentation or push for it.
- Scalpel Blade: Cut the inside of the hula hoop all the way round. Messy and only done in third world.
The non invasive procedures:
- SLT laser (selective laser trabeculoplasty) Don't know much about this and could be done in a clinic. Tries to help heal the trabecular structure. (inside of hula hoop) 5-10 minute procedure.
The things that may move the dial from a sales perspective:
My surgeon did not seem to want to use it anytime soon. His offhand remark was "may be useful in the third world if its cheaper" However the way the US is structured for reimbursement with MIG surgery at the moment that may well fit.
Australia:
- Comes down to how the ophthalmologists are trained in their fellowships in UK and Canada. So a question to the company about the people who are using it there might be an idea. That will feed back into Australia and put pressure on the reimbursement system with the more who use it. This is a long term process. However not even sure it could compete with the hydrus stent on a long term basis. All I can see on their website is a 3 year study vs the 5 year study for hydrus (haven't had a solid look). Also hydrus is reimbursed here, but the gap that is charged by my surgeon is only 100 dollars extra.
- Time it takes to put in? Not sure for Itrack. A cataract and hydrus stent is about 20-25 minutes combined, including anaesthetic time.
US:
- According to the Alcon rep: MIG surgery reimbursement was decreased to a measly 200 dollars in 2020. So there is no incentive for the US system.
- Not sure what the sales process in the US is doing (I'm sure this was answered in the presentation)
Just trying to understand where it fits in the ecosystem of treatments. Very possible to go from the 3 to 7% target for that 60million revenue. I just don't think that it is necessarily a better treatment so will need to play the incentive card with prices and legwork on the ground +/- change the reimbursement. If they have a smart way to do that, they could well succeed very nicely. But all this takes time and money and in device world, if they don't have a long runway of cash may be a problem. I'll have a listen to the meeting and no doubt that will enlighten me on the business side of things a little more there.
Cheers,
Note: Pathway of aqueous humor:
Produced in ciliary body --> flows through the posterior chamber of eye --> between the iris and the lens --> into the anterior chamber of the eye --> through the trabecular meshwork --> into canal of schlemm. (THIS IS THE TREATMENT SITE), through small and large channels (important for understandind difference between Istents and hydrus stents) and back into the blood flow
mikebrisy
@Metis I hear you! Perhaps it is all a manifestation of the fabled saying ... "In the land of the blind, the one-eyed man is king!"
There is no question that there is a world of difference between the US and other markets, driven both by i) regulator behaviour, ii) reimbursement regimes, iii) market structure (including privatised vs. single-buyer-type public medicine), iv) established standards of care and professional consensus.
This has shown up in pretty much every area of pharma or devices I have investigated over the years - particularly where we are talking about innovative and/or "me too" products.
As I have researched the clinical evidence around glaucoma, several things have occurred to me.
First, the ultimate progression of the disease is relatively slow (usually) spanning many years or decades.
Second, even the common endpoint - IOP suppression - is subject to differences in measures (washout vs non washout). This, combined with the relatively large overlap in therapeutic effect compared with the mesurement confidence intervals, and disease state variations in the underlying populations, makes is hard to support claims of relative efficacy of one technology over another. To one your original challenge, I think this explains why $EYE haven't invested in a true head-to-head randomised controlled trial of their product to another MIGS product. In seems their commitment to the iTrack Register is the best we will get. As Tom said, he can get a greater return on investment by adding US reps than by investing to try and achieve Australian reiumbursement.
Third, relative to the disease progression, many treatments are relatively new to the market. So, for example, the first MIGs (iStent) received FDA approval only in 2012 (I think), so MIGS have only been around for 14 years, and many of the products for a significantly shorter period of time. This means that the timeframe to monitor the impact of each treatment on disease progression has been relatively short.
One illustration is the first line treatment for mild/moderate early stage glaucoma which is the laser treatment - SLT - which is increasingly being favoured in Australia by ophtlamologists over drops (e.g., better quality of life).
The LiGHT trial (Laser in Glaucoma and Ocular Hypertension Trial), led out of the UK by Moorfields Eye Hospital and UCL, compared first-line selective laser trabeculoplasty (SLT) with initial treatment using pressure-lowering eye drops in newly diagnosed open-angle glaucoma or ocular hypertension.
The 3-year results (2019) showed SLT achieved equivalent intraocular pressure control, with around three-quarters of patients remaining drop-free, fewer surgeries, and superior cost-effectiveness. The 6-year extension confirmed durability, with roughly 70% of SLT-first patients still not requiring drops and lower rates of disease progression and incisional surgery. Longer-term follow-up (reported at ~10 years) reinforced sustained pressure control, continued reduction in surgical intervention, and maintained safety, supporting a structural shift toward SLT as a viable first-line therapy in early glaucoma.
When investments like this are made, the payoff is huge. How did I find out about it? In my chat on Monday with my Dr, when I asked why he thought there was better or more comparable data available.
Coming back to $EYE, with a 10 year, reputable and ongoing study for SLT, that raises a pretty high barrier that iTrack would have to clear for wider adoption. I mean, we know it is likely to give a comparable result to OMNI in pure canaloplasty mode, and no matter how good the result, SLT has a 10-year dataset.
So even if iTrack got reimbursement in Australia, it seems to me that it addressible market might only be the 25% of the market that appear not to have such a great response to SLT (which apparently can be measured 6-9 months down the track, when IOP comes back quite quickly.)
So the next question is, where dies SLT sit in the US? How is the LiGHT data being viewed in the US?
So finally I get to my major insights (sorry for being long-winded but it might be worth it)
It appears that the US market has evolved with drops as the first line of treatment. Through the 1990s and 2000s, new products were introduced, including combination therapies.
In the 2010s MIGS (stents) started to make inroads into the market, as the least invasive surgical intervention, often called up when drops failed.
While SLT was introduced into the US, clincal practice had drops, then MIGS entrenched,
As MIGS gained market penetration, innovations like OMNI and now iTrack, are targeted on earlier interventions, which leave no hardware behind and are less invasive from the perspective of tssue damage.
Until a few years ago, it seems that it was common practice for higher value Stent codes (66175) to be used for canaloplasty_trabeculotomy, until CMS started clamping down and now canaloplasty and canaloplasty+trabeculotomy have to be filed in the less lucrative (66174).
The ecomonic incentive really kicks in for the combined cataract and canaloplasty surgery. Here, for 30 minutes of clinic time (cataract, US$550 per eye), the surgeon can get an incremental US$540 per eye for only an incremental 6-15 minutes of work. The economics of the combined procedure are compelling.
So, how will LiGHT evidence impact this?
It is unclear at this stage how the US industry is responding to the evidence that shows that both form an efficacy perspective and also quality of life. SLT looks like for 75% of the population it is the best first line treatment for early detection of mild/moderate glaucome.
In the US this procedute attracts a Medicare Physician fee of $240-$350 per (eye). Total chair time is 10-15 minutes, and laser time is about 5 minutes. The treatment is typically performed over two visits, doing 50% of each eye per visit, and a follow-up exam.
Because the laser is a capital item, there isn't large device cost, and so the Facility Fee is relatively small. You can understand why when you look at the economics of investing in an SLT laser (US$40k up to US$80K):
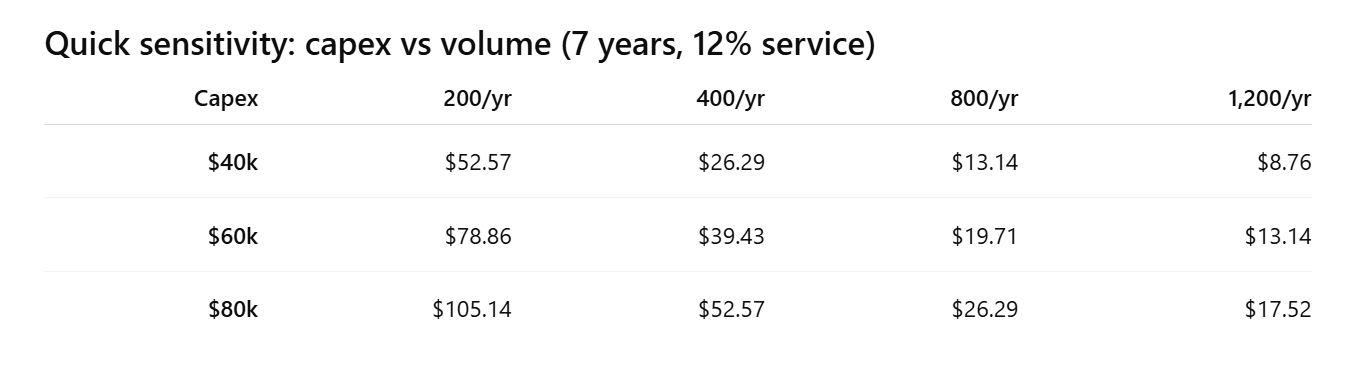
My Conclusion for the Investment Thesis
There is not a lot to go on as far as clinical evidence is concerned.
Of the MIGS procedures, iTrack main selling point is a minor edge on safety.
In the US, the economics incentives for the physician at current reimbursements level, appears to favour a combined cataract + canaloplasty procedure. And I think this explains why the simple iTrack device aligned with this combination procedure, is getting traction in the US.
However, there is a real risk that the US starts to respond to the SiGHT 10-year data, which is now (as far as I can tell) more compelling and complete than data for canaloplasty and trabeculotomy.
However, the entrenched US practice is eye drops, then MIGS, and the economic incentives favour combining cataract + MIGS to get patients off drops.
From a total cost of treatment perspective, my quick reading on the healthcare economics is that it favours SLT.
If this is true, we could see further adjustments in CMS codes to incentivise the industry to move more towards SLT, as the first line.
Until I better understand that risk, I don't think I can invest in $EYE.
There's a lot I don't know, ... but I know a hell of a lot more than I did one week ago.
Looking forward to this afternoon's meeting!
Critique of My Work
I have been too simplistic here. For example, SLT adoption in the US might simply expand early treatment, and not compete with the combined Cataract + MIGS pairing.
The economic incentive to couple cataract + MIGS appears strong, and US clinical practice appears established. This could be hard to break.
However, one thing that I think is clear ... there is a MAJOR barrier for iTrack to get reimbursement in Australia (and similar markets), and I think it is clear $EYE have put that in the too hard bucket.
Looking forward to tonight's discussion.
mikebrisy
OK, at the risk of overloading, I want to try and tie all my insights back to the US Market opportunity. Here goes.
(This analysis has been assisted by my BA, but they sometimes need help unzipping all the CMS datafiles, so I'll credit myself as co-analyst)
It has finally dawned on me how canaloplasty+trabulectomy (OMNI) and now pure canaloplasty (OMNI + iTrack) have been marking inroads into the US market for early and mild/moderate glaucoma treatment.
Consider the following story, viewing from the physican's and also patient's perspectives
In the US, most ophthalmologists begin as cataract surgeons and manage mild glaucoma with drops or selective laser trabeculoplasty (SLT) in clinic.
SLT is commonly offered early because it is quick, repeatable, and avoids implants. As surgeons gain comfort operating in the anterior chamber angle (through fellowship training or incremental MIGS exposure) some begin adding minimally invasive glaucoma surgery (MIGS) at the time of cataract surgery, particularly for patients with mild–moderate glaucoma already scheduled for the operating room. In this context, MIGS is typically framed as a medication-reducing adjunct rather than a replacement for SLT. (That's a new insight I didn't have in my prior post.)
Stents are the usual entry point. They have been established longer, so there is a wider support base of clincal mentors and sales rep penetration. Placing a stent is also a less technically challenging procedure than a canaloplasty/trabulectomy, particularly for an experienced cataract surgeon.
Of the some 9,000 US opthalmic surgeons who perform cataract procedures, anywhere from 2,700 to 4,000 have performed any kind of MIGS. And stent-base procedures were most likely their entry point. Of course, many provide SLT procedures too, and some may stick to cataracts and SLT alone.
Over time, adoption often progresses from focal trabecular stents toward broader angle-based procedures. Increased interest in canaloplasty reflects that it dilates the full 360° of Schlemm’s canal, leaves no permanent implant, fits efficiently into cataract workflow, and aligns cleanly with current reimbursement structures. Many surgeons therefore offer both SLT (in clinic) and MIGS (in the OR), with canaloplasty positioned as a no-implant, cataract-adjacent option rather than a direct substitute for laser therapy. (My earlier post questions whether this will change in the light of unfolding clinical evidence? I haven't seen any clinical data yet for SLT + MIGS outcomes.)
From a physician perspective, canaloplasty can be attractive because it avoids implant inventory, tracking, and potential device-related complications, while still addressing outflow resistance more diffusely than a focal stent. "Tracking" means that as a higher cost procedure, with a history of some misuse, there is increased auditing of use of stents by CMS, so potentially a perceived administrative burden (although I find this hard to really swallow given the integration of IT systems into clincal and surgical workflow).
Compared with combined canaloplasty plus trabeculotomy (e.g. OMNI), standalone canaloplasty (OMNI and iTrack) may be perceived as less aggressive, with potentially lower bleeding risk and a more conservative safety profile — suitable for earlier-stage disease. Indeed, the one clinical claim that iTrack appears to have over OMNI is less transient bleeding, which may be a consideration for what is a walk-in-walk-out day surgery.
From a patient perspective, the appeal also often lies in “no hardware left behind,” the possibility of reducing drops, and adding the procedure during already-planned cataract surgery without meaningfully extending recovery. So, lose the drops without any extra effort!
Together, these clinical, economic, and psychological factors are contributing to steady uptake of canaloplasty within the cataract-MIGS segment rather than displacing either SLT in clinic or more invasive glaucoma surgery in advanced disease.
Implications for $EYE's US Market Positioning
CEO Tom Spurling has confirmed my research that the market sweetspot is targeting the general opthalmic surgeons who are doing cataract surgery and also treating glaucoma.
Those who appear most susceptible to iTrack adoption appear to be either the 2,700-4,000 who are already providing MIGS procedures, and possibly targeting what I estimate to be less than 1,000 who are already doing canaloplasty (likely using OMNI).
If they are focusing on this smaller segment, this explains why they have such a small sales force of c. 10 FTE. A ratio of 100:1 clinicians to sales reps is at the high end and, at this level, it is almost impossible they $EYE are addressing their prirority target market of the 2,700 - 4000 surgeons already performing MIGS and they are geographically constrained.
Clearly, where there are geographical overlaps, a rep will work on both an "OMNI Surgeon" to convey the key messages of reduced bleeding risk, as well as a "Stent Surgeon" to sell the workflow and patient perception advantages of canaloplasty, while probably making the point that canaloplasty alone using iTrack is likely good enough, and there is no need to use the more flexible OMNI product.
OMNI's reveues are holding roughly flat, likely because the flow of new surgeons from stents to OMNI, is being matched by the switching of surgeons from OMNI to iTrack.
Clearly, CEO Tom is laser focsued on getting to positive operating cash flow, and even though he is achieving $2million revenue per US rep (which is way above the benchmark in this space), the small number of reps MUST MEAN that he is accessing only the market sweet spot.
The "spreadsheet" that he talks about probably lists both the current accounts and potential account in each major urban cluster.
Here's how I would deploy 10 reps: 2 Northeast Corridor, 2 Socal, 1 Texas triangle, 1 South Florida, 1 Great Lakes, 1 Ohio Valley, 1 North Cal, 1 lots of choices!!
That likely leaves each rep under-serving their "catchment" and A LOT of the US urban geography uncovered - they are probably using conferences to try and pick up some of these.
I believe that once $EYE become cash positive (i.e. in FY26 at the operating level), over time, Tom can easily DOUBLE the sales force to get a more complete coverage of the major urban centres, and probably infill the NorthEast Corridor and SoCal to 3 reps, easily.
That will enable him to more aggressively tackle the 2,700-4,000 Ophths who are already doing combined cataract surgery and either using Stents or OMNI.
Conclusion
As I crunch the numbers, this actually looks pretty positive.
There is enough evidence that US surgeons are inclined towards device-based surgery, and that canaloplasty is gaining traction, and that $EYE is under-resourcing the market opportunity. Despite holding salesforce FTE flat and at a level way below benchmarks, $EYE delivered 32% US revenue growth in FY25 over FY24. That indicates serious momentum, and gives me a good basis for some valuation assumptions. (The next task).
My remaining questions are:
- How sticky is OMNI is once adopted?
- Whether surgeons prefer dual functionality (canaloplasty + trabeculotomy)? (SIGHT SCIENCES flat revenue indicates "No")
- Whether bleeding differences are commercially meaningful?
- Whether stents rebound with next-gen data and what about SLT with growing evidence?
- Whether reimbursement shifts again?
Metis
@mikebrisy your ability to hit some solid points so quickly is pretty damn epic mate.
It makes logical sense that it would replace Omni due to the decreased trauma to the eye. Potentially less debris/bleeding and transient visual issues post. However I guess there will be warring studies.
It makes sense to target those cataract surgeons in the grey area of needing to deal with mild glaucoma whilst doing a cataract who are not glaucoma sub specialised. that is a large subset to sell to…
thanks for the replies. I’ll try and make the meeting pending work.