Price: AUD$0.60; Shares Outstanding: 334.9 million; Market Cap: NZD$249m (AUD$208m); Enterprise Value: NZD$226m (AUD$189m)
All monetary figures unless otherwise specified are in NZD
For a detailed background on this company, I highly recommend reading Mondy’s fantastic writeup on Substack: https://mondyinvest.substack.com/p/long-idea-aroa-biosurgery-asx-arx
Quick and dirty company background
Aroa Biosurgery is a commercial-stage soft issue regenerated company headquartered in Auckland, New Zealand. The company was founded in 2008 by Dr Brian Ward, who continues to serve the company today as CEO and Managing Director, owning ~10% of the shares outstanding. It listed on the ASX in mid-2020, raising $45m at $0.75/share and quickly shot up to $1.35/share on the first day of trading.
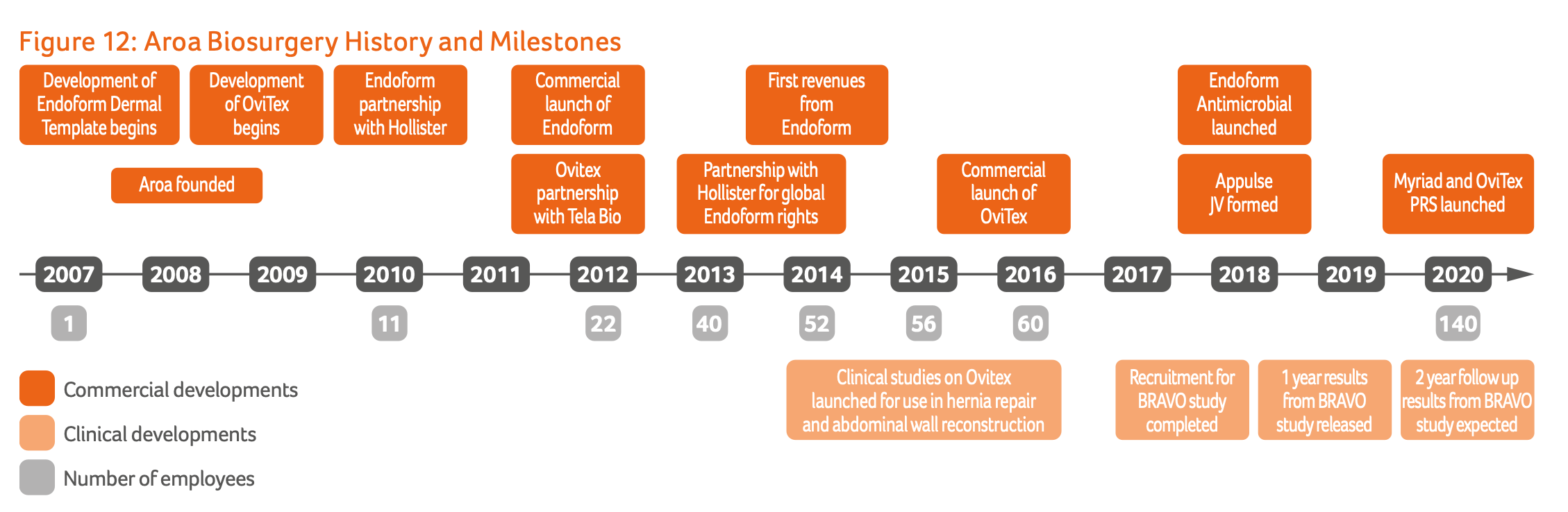
All of Aroa's products are built on the Aroa ECM (Extracellular Matrix) Platform, which is derived from the forestomach tissue of New Zealand sheep. The forestomach is a rapidly growing and regenerating organ with a unique structure optimised for nutrient absorption. Aroa's proprietary manufacturing process removes cells to prevent immune rejection while preserving the tissue's biological structure and bioactive molecules. The result is a biological scaffold that retains native vascular channels, enabling rapid blood supply establishment and kickstarting the body's natural tissue regeneration process.

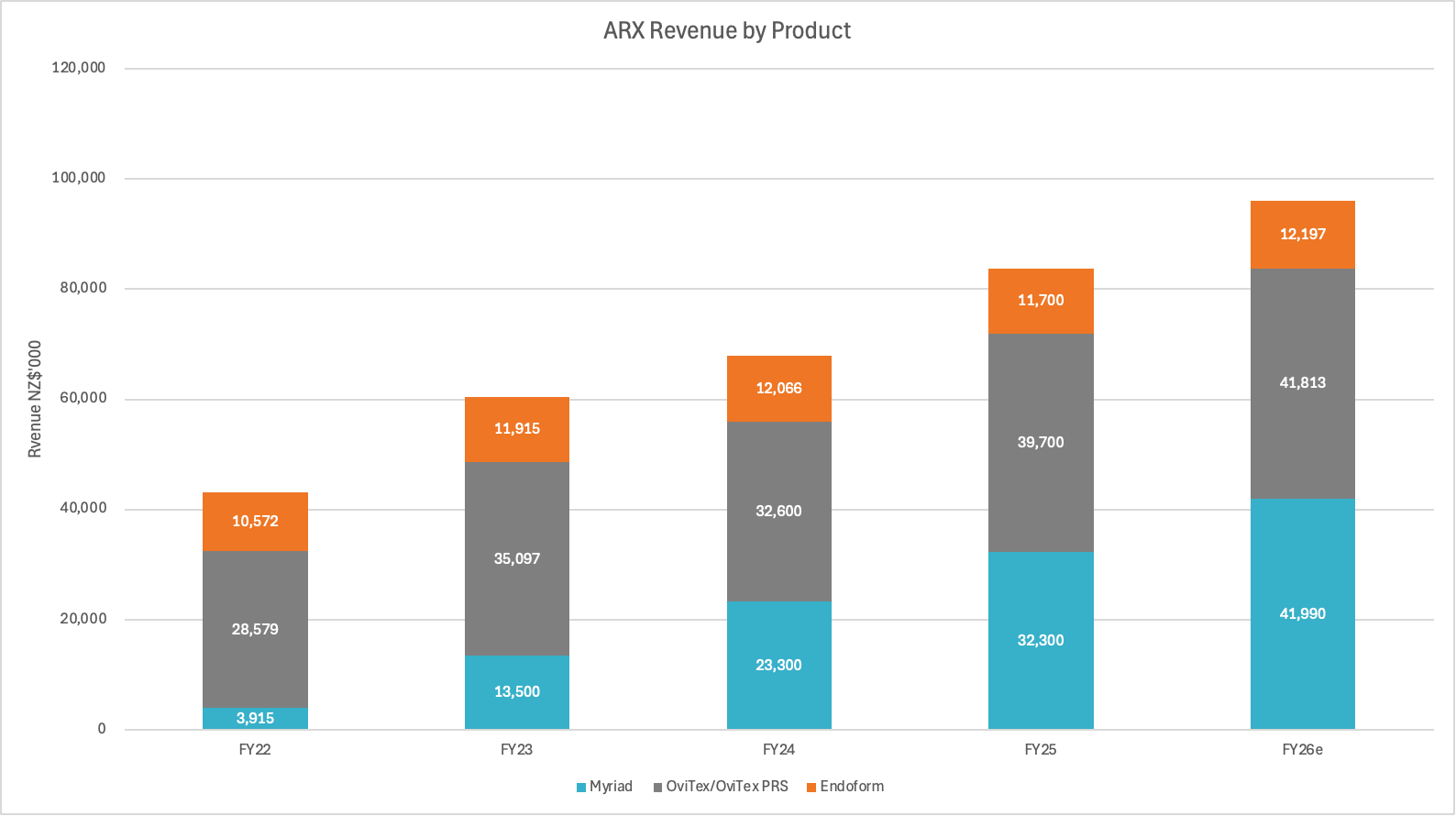
Today, the company is a manufacturer of 3 key products - Myriad, Ovitex and Endoform - with 95% of its revenue coming from the USA. The company is highly dependent on its sales partner TELA Bio to distribute its hernia and breast reconstruction products - TELA handles all the sales, marketing and distribution around these products.

Why is it interesting?
Good Division / Bad Division
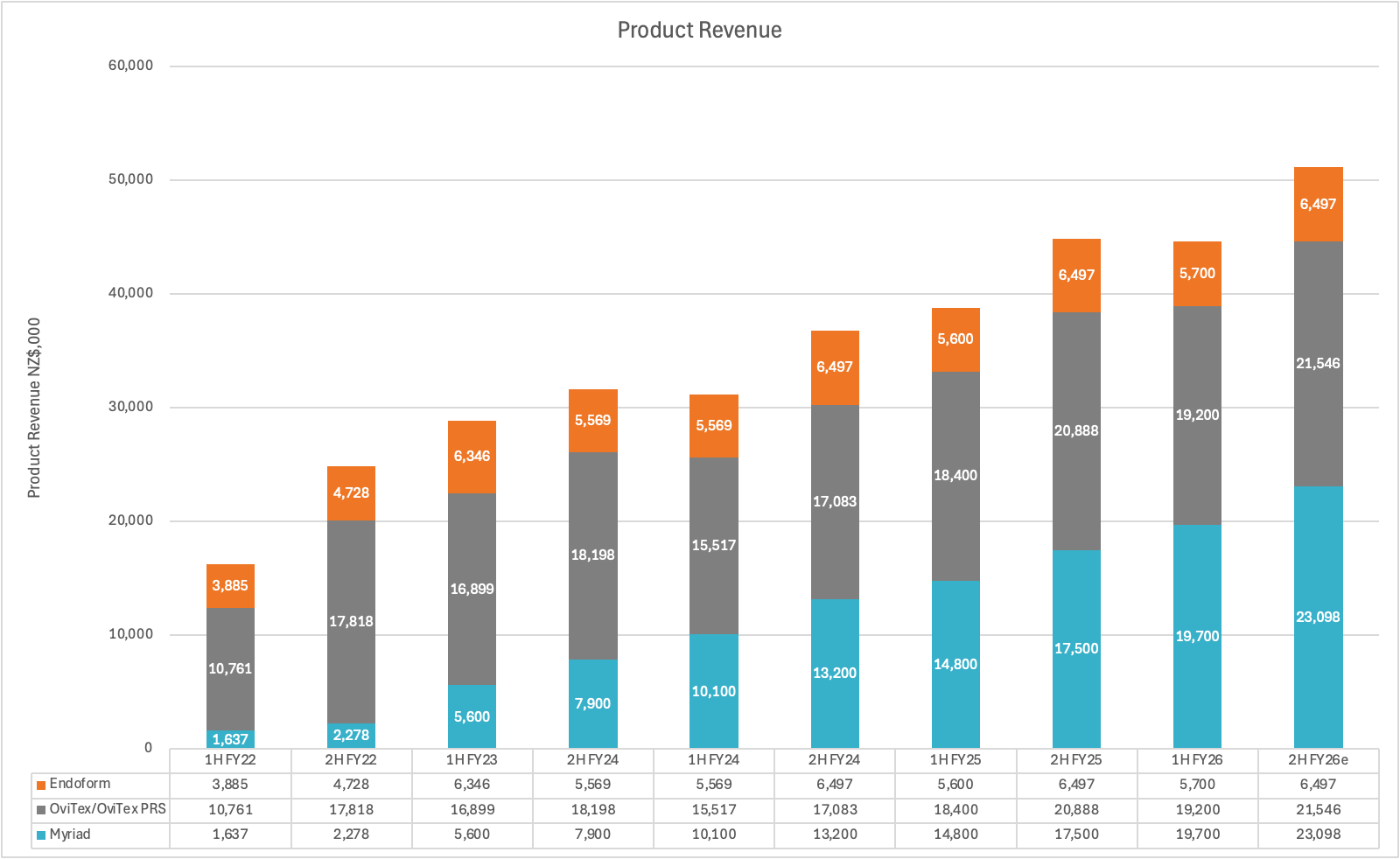
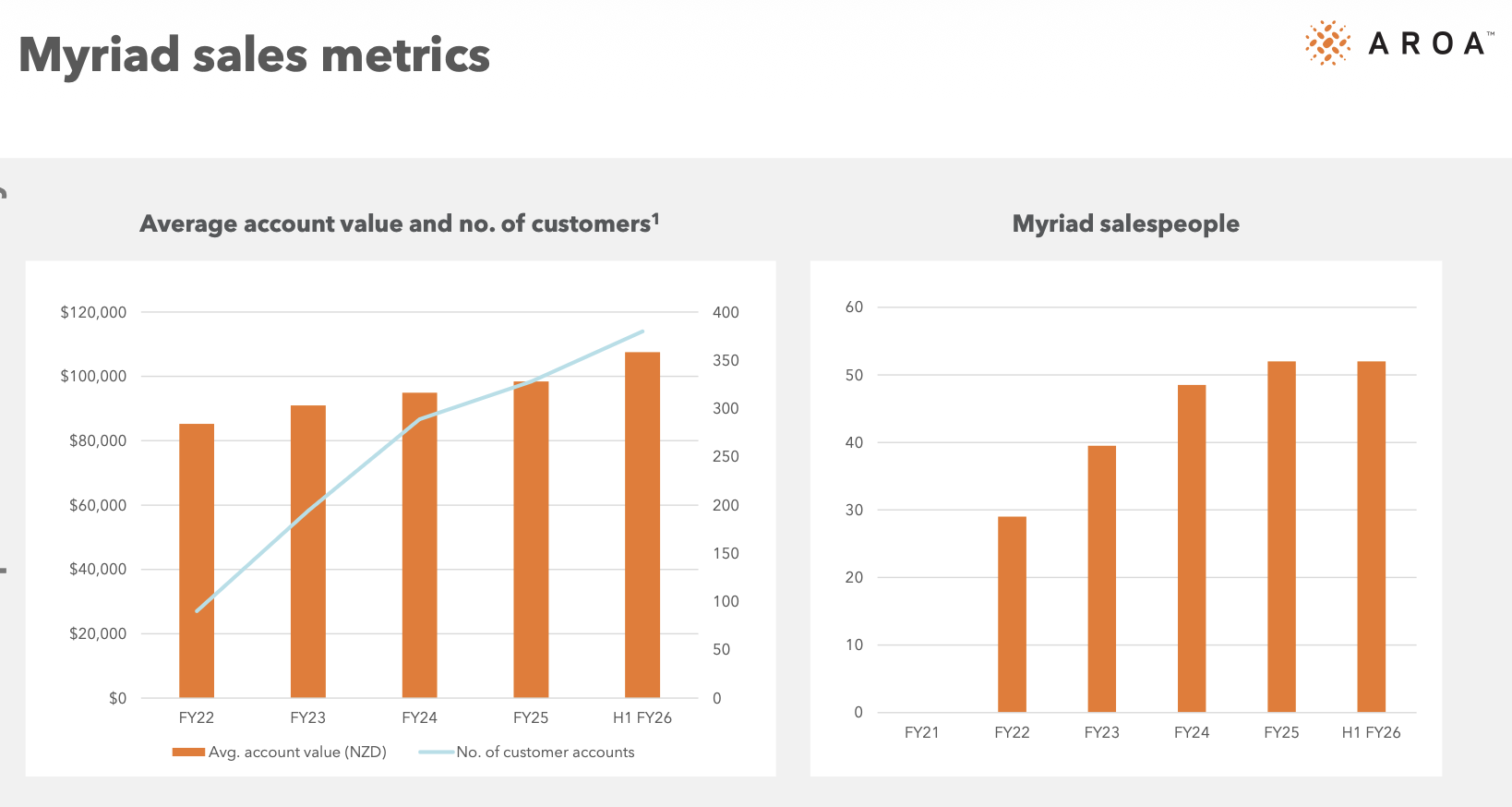
The top line growth rate over the past 3 years has been strong - revenue grew from $60.5m in FY23 to $96m in FY26e at a CAGR of 16.6%pa. However in that period, OviTex only grew at 5.3%pa as TELA Bio struggled with monopolistic competitors and Endoform flatlined (mature product). The true growth engine has been Myriad which was only launched in FY22 and now account for 45% of revenue. It’s now the company’s largest product line, is still growing at circa 35%pa and will make a much larger contribution to the company’s top line growth going forward.
Myriad shows no signs of slowing down, with both account numbers and average account values continuing to increase. Meanwhile, sales staff growth has slowed over the last couple of years, demonstrating emerging operating leverage. Management believes the soft tissue reconstruction market is US$730m in size, with Aroa's share at only US$28m annually, suggesting plenty of room for continued growth.
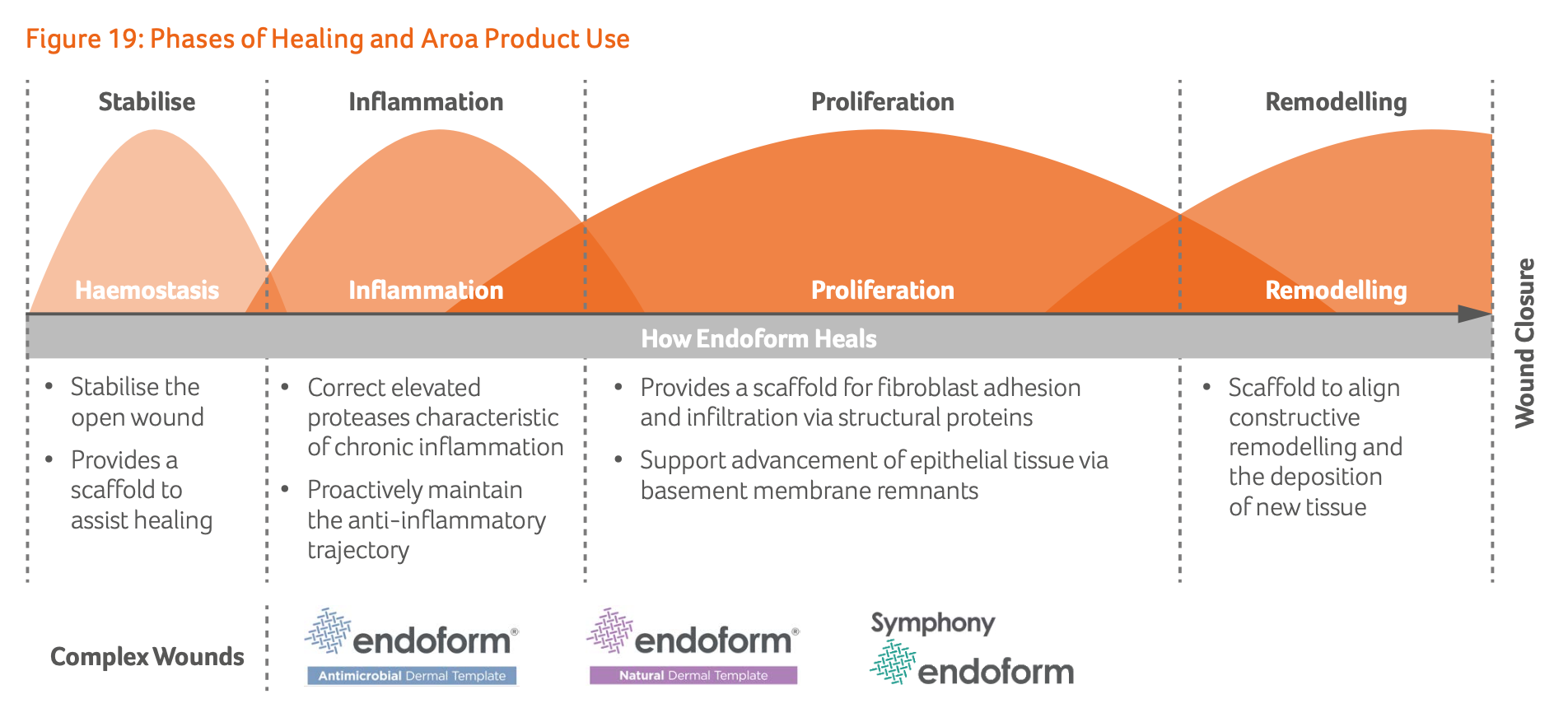
Symphony: Another leg to the stool?
Aroa’s Symphony is a product that is applied on complex wounds during the proliferation phase of wound healing and competes in the CTP (Cellular and/or Tissue-based Products) market, commonly referred to as "Skin Substitutes.” The product gained FDA clearance in 2020 and was launched commercially in 2023, but has been kept in a holding pattern since.
Symphony was placed on hold due to a dysfunctional CTP outpatient reimbursement market. Essentially, skin substitute manufacturers and doctors were collaborating to exploit Medicare by charging artificially inflated prices (e.g., US$2,000/cm²) and pocketing large commissions. Aroa, being the high integrity company that they are, only charged US$70-100/cm^2, and refused to participate in the scheme. As a result, the product never gained traction. You can read more about it here: https://oig.hhs.gov/reports/all/2025/medicare-part-b-payment-trends-for-skin-substitutes-raise-major-concerns-about-fraud-waste-and-abuse/
However, on January 1, 2026, new Medicare payment rules took effect to close the loophole by establishing a single outpatient reimbursement rate for skin substitutes at $127.14/cm². This has created a much more favourable environment for Symphony, as it's likely to push out competitors with expensive products and costly distribution models. The company intends to ramp up sales starting in FY27.
To directly quote the CEO from the last conference call: “I think we would expect Symphony to at least have the same sort of growth trajectory as we've seen with Myriad previously.”
Myriad generated $3.9m in revenue in its first year and $13.5m in its second. If Symphony replicates this success, it would add another major product line to the group, creating an additional growth engine for the business. Symphony is also complementary to Endoform, as together they provide products covering the entire wound healing lifecycle, which could also boost sales of the latter.
Yesterday, the company announced that the Symphony Randomised Controlled Trial (RCT) is now complete and preliminary findings indicate that the study achieved the primary endpoint. Once the results are confirmed and published, it should further support the use of Symphony for the treatment of non-healing Diabetic Foot Ulcers (DFUs) and any future changes in reimbursement policy.
Profitability inflection
Over the past few years, Aroa has been investing heavily in building out its own sales team to support Myriad's growth, expanding its manufacturing capacity, and developing new products (including ENVIO for a estimated 2028 release). The company has now reached a profitability inflection point where incremental sales and marketing expenses and R&D spend are moderating, allowing earnings to turn positive. With the Symphony randomised clinical trial having just concluded, I expect expenses as a percentage of revenue to continue to moderate in FY27 as the jaws of operating leverage open wider.
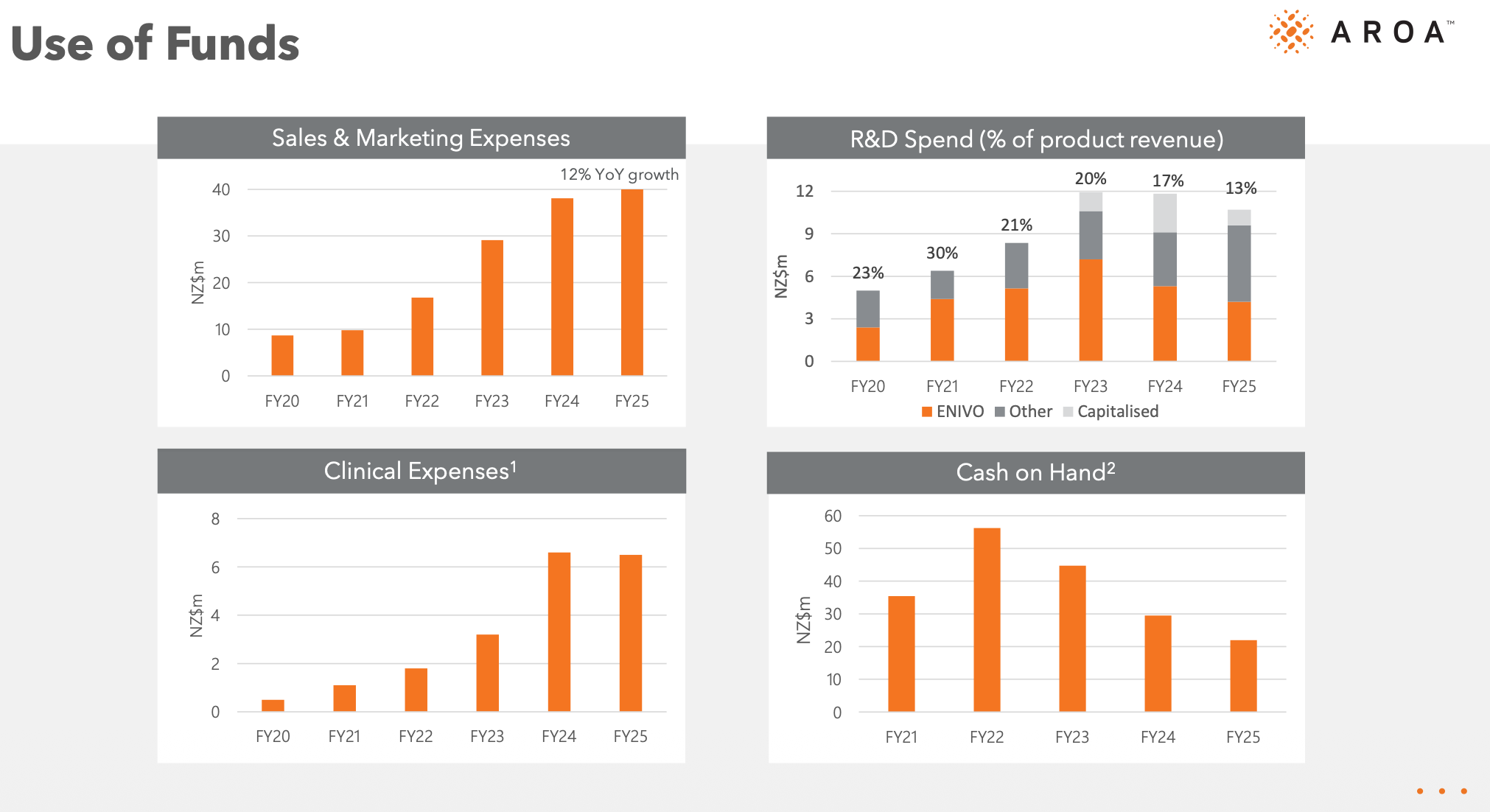
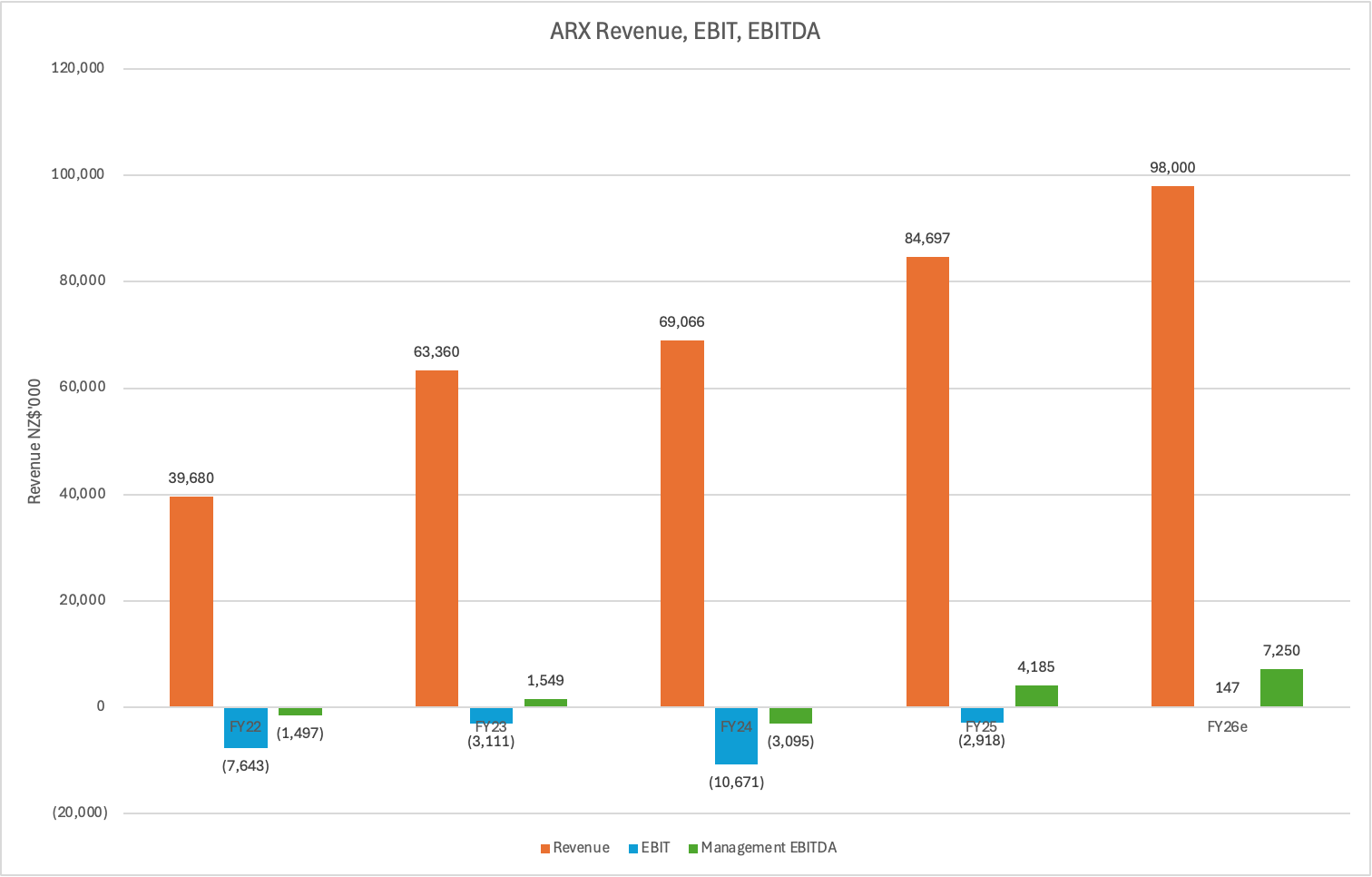
With top line growing at 15%+ and expected to accelerated, 85% gross margins, inflecting into profitability with signs of operating leverage evident - all the while trading at 2.15x FY26 revenue. It’s definitely not expensive for what appears to be a quality and growing business.
With top line growing at 15%+ and expected to accelerated, 85% gross margins, inflecting into profitability with signs of operating leverage evident - all the while trading at 2.15x FY26 revenue. It’s definitely not expensive for what appears to be a quality and growing business.
Risks
TELA Bio
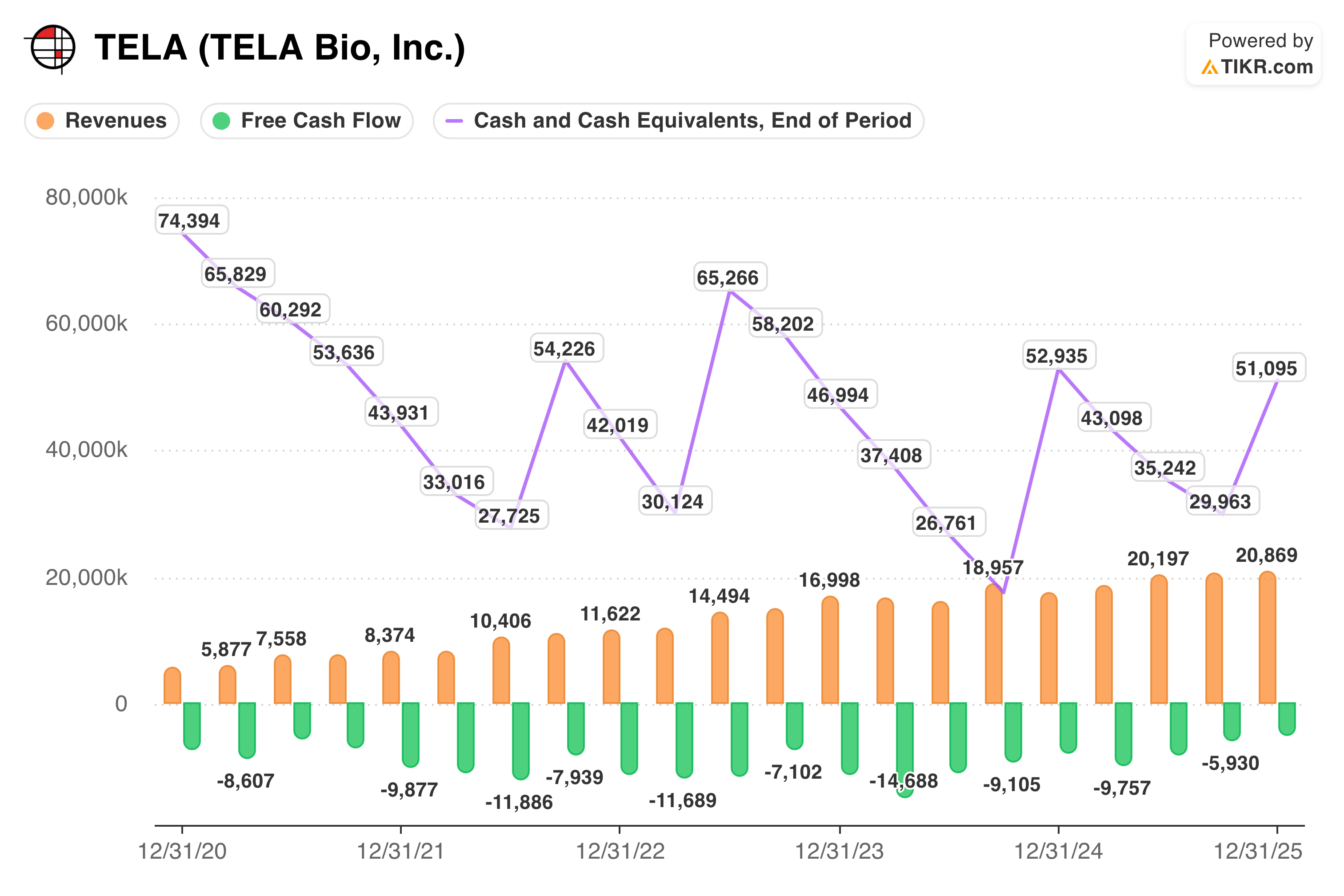
TELA Bio is Aroa's commercial partner in North America and Europe for its OviTex branded products, which are used in hernia repair, abdominal wall reconstruction, and breast reconstruction. Under this partnership agreement, Aroa handles manufacturing and product development while TELA Bio manages sales, marketing, distribution, and clinical trials. In return for its contribution, Aroa receives 27% of TELA Bio's net sales from these products.
This arrangement currently represents approximately 43% of Aroa's total revenue and generates highly attractive economics - 70%+ gross margins with strong earnings margins thanks to minimal costs below the gross profit line.
Despite strong revenue growth, TELA Bio has never achieved financial sustainability, burning substantial cash each quarter. The company has survived through periodic capital raises and debt financing. Most recently, in November, they raised an additional US$11.9m in equity and increased their loan facility by US$14.1m, with an option to extend another US$10m. With US$41.8m in cash on hand and undrawn debt capacity, the company has roughly two years of runway at its current burn rate, assuming no significant improvement in the underlying business.
TELA Bio's financial struggles pose a major risk to Aroa. If TELA Bio runs into serious trouble, it could disrupt sales and hurt Aroa's revenue. However Aroa’s position has been that the OviTex portfolio is still a US$80m sales/year business that is very valuable, and who’s economics would be attractive to larger industry players who could acquire the assets and operate them more efficiently.
OviTex's share of Aroa's revenue is declining significantly - from 73% in FY22 to 43% currently, and expected to fall to approximately 30% within two years. The longer TELA Bio can maintain operations in its current state, the less disruptive any potential interruption would be, as Aroa continues to grow and diversify its product portfolio beyond its dependence of the TELA Bio partnership.
Myriad competition
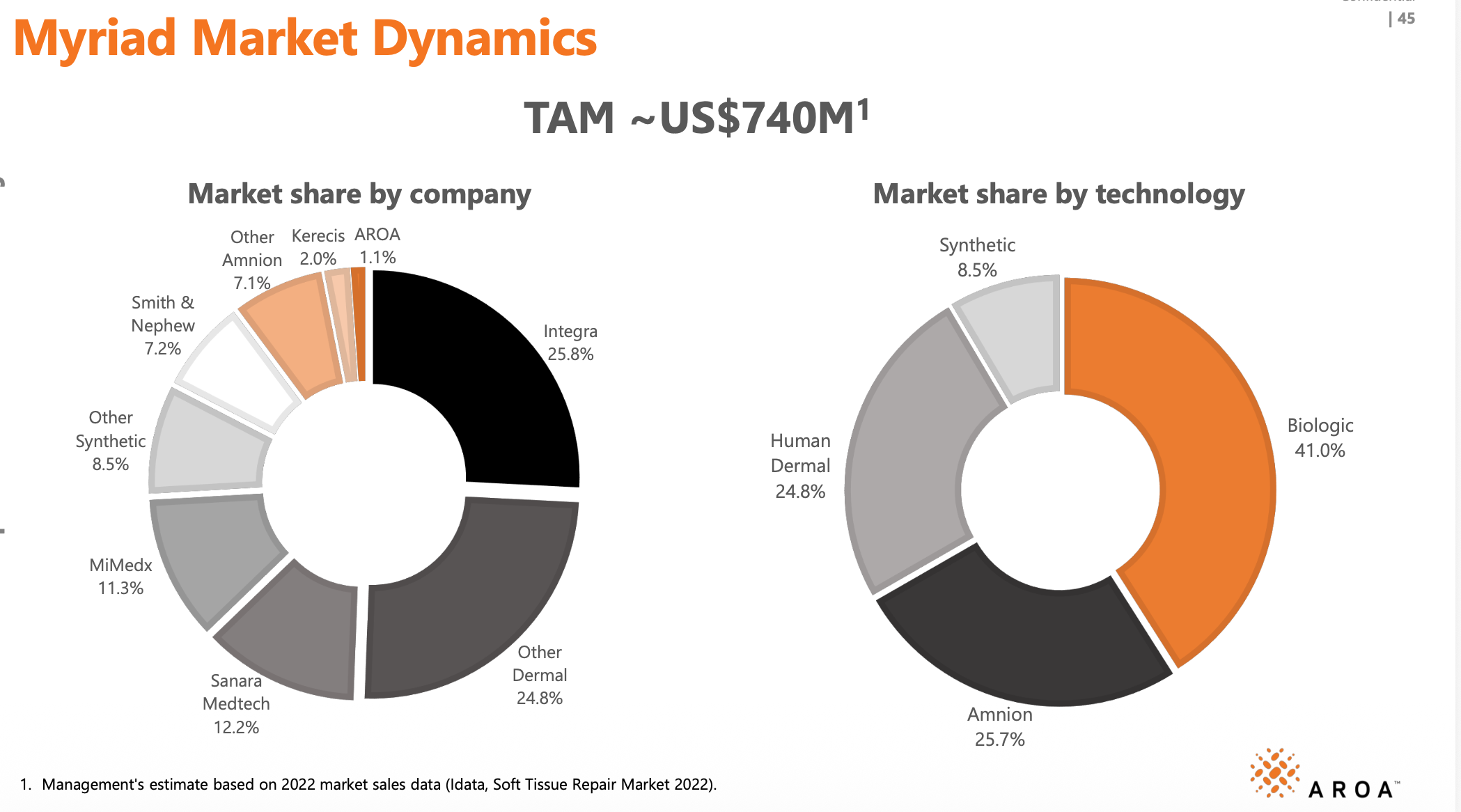 Note: Myriad now has~3.8% market share
Note: Myriad now has~3.8% market share
Aroa’s Myriad is positioned as a premium biologics product, with all the superior healing properties, while carrying a significantly lower price point and very low rates of inflection and complications. That said, the soft tissue reconstruction market is a fiercely competitive one.
One watch point is the growing traction of synthetic solutions within the space. While synthetics cannot match biologics on healing outcomes or complication rates, they are winning on three fronts: cost, consistency, and infection resistance. Newer products such as Polynovo's Novosorb MTX are also improving and addressing historical disadvantages of synthetics such as the need for multiple applications or two-stage surgery - further narrowing the gap.
Another watch point is Integra's PriMatrix, a like-for-like alternative to Myriad, which is returning to market after an extended absence. Integra encountered serious contamination issues at its Boston facility in mid-2023, leading to the suspension of several products, PriMatrix among them. The product relaunched in Q3 2025 using third-party suppliers, though Aroa has yet to feel any impact, with management having recently upgraded FY26 guidance, implying continued strength in Myriad sales.
Conclusion
Aroa Biosurgery is a fast growing, high-margin wound care company that is just beginning to inflect into profitability.
The growth rates are somewhat understated due to mature and stagnant product lines that previously dominated the company’s revenues. Myriad is Aroa’s key growth driver, and having only launched in FY22 is now the company’s largest product by revenue. Its substantial annual growth rates of circa 35% should now begin to impact revenue and lift growth rates in a more substantial way at the group level.
And with Medicare payment rules having recently changed significantly, the time is now right to re-launch Symphony - a product management believes should re-enact the success of Myriad. This could very well create a second major growth driver for the company in the coming years.
The company’s year end is 31 March and the FY26 report is expected by the end of April 2026.
Disclosure: At the time of writing I hold shares in ASX:ARX

















 Note: Myriad now has~3.8% market share
Note: Myriad now has~3.8% market share