Some Background - Why Am I Even Bothering?
(Perhaps against my better judgement and experience) last year I initiated a small position in $MVP (of Green Whistle fame), given some indications that good growth continues in Australia, and the impending European pediatric indication (August 2025 HPRA extension approval for children > 6 years) bodes well for growth in Europe, given that having a combined adult and pediatric indication increases the likelihood of adoption in emergency rooms and ambulances/paramedics (Evidenced by one NHS Trust already).
Having struggled through the pandemic for obvious reasons, having abandoned its attempt to enter the US (legacy of historical reputational overhang of toxicity in previous high-dose, anaesthsia applications in the 1960s and 1970s), having upgraded management and board with more internationally credentialled people (bye bye DW), a number of historical negative flags have been taken down, and so I now hold an initial 1% position (RL) having avoided the business for the last 8-9 years.
I'm still not convinced it is a good move, and will await a couple more quarterly reports before deciding to add or drop. That's because I need to see if Australian momentum can be sustained, and if UK/Europe usage ticks up. If that happens, then the business could start to look attractive. It is currently on an undemanding FY26 revenue multiple of 0.8x.
Today's report is not bad. I'm not going to get too excited about positive operating cashflow for the half, as there is varaibility from period to period, and we need to see Europe come to the party and start driving stronger growth than the somewhat anaemic +10%, given low current adoption. At the first sign that this is happening, I'll add.
Their Highlights
• Positive cashflow from operating activities for FY26 half year.
• Penthrox revenue up $2.3 million.
• Penthrox volume growth of 26% in the Australian hospital segment.
• Penthrox PBS Prescriber Bag eligibility extended to Nurse Practitioners in Australia.
• European in-market Penthrox volume growth of 10%.
• MAGPIE paediatric study published in Injury.
• Approvals for the Penthrox paediatric label in Europe progressing to plan.
• Cash balance at 31 December 2025 of $16.9 million.
My Analysis
Here's my usual 4C trend analysis over the last 8Q.
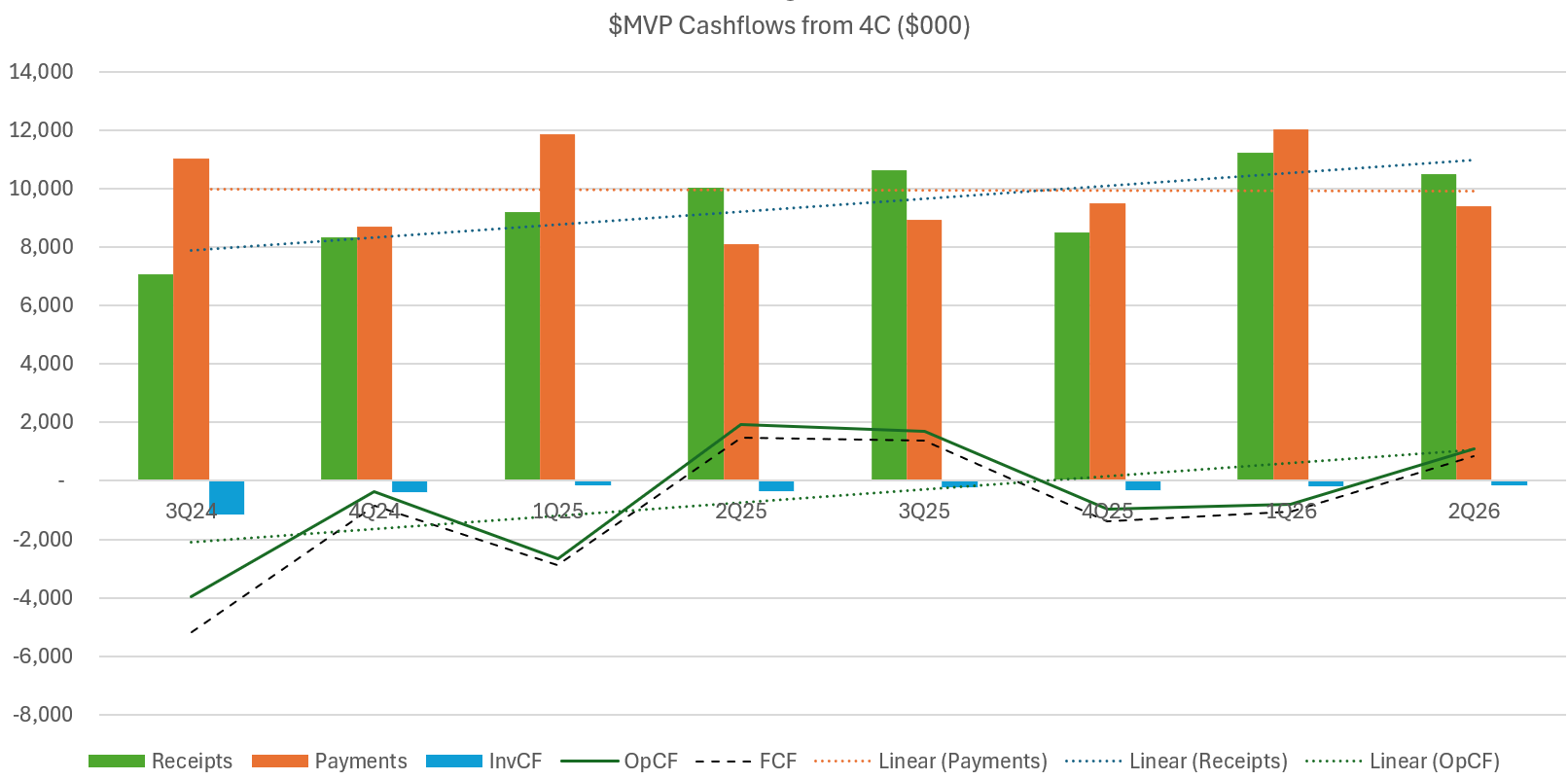
Costs were well controlled, albeit receipts weaker than trend, driven by softer revenue in the last Q.
The 8Q trend indicates that the business is moving into overall cash generation, and so the question is can we see uplift in Europe over the coming years?
There's good volume growth in Australian hospitals to provide a robust revenue base, and the extension to Nurse Practitioners in Australia, while unlikely to be material, add some support. (There are about 2,250 nurse practitioners in Australia, a tiny part of the c/ 138,000 registered clincal prescribers. But they could be an important first line users in remote and regional areas.) Rather than being major news in its own right, it is part of a continued pattern of wider adoption of the product.
Conclusion
An OK result, neither thesis confirming nor destroying. I am prepared to monitor while holding a small position over the next few quarters, unless the capital is called away elsewhere (which is entirely possible!)
Disc: Held (RL 1%, but low on conviction list)



